Download The EIRENE Code User Manual Version: 11/2009
Transcript
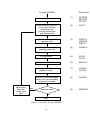
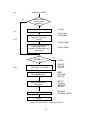
* NEUTRAL MOLECULES SPECIES CARDS: NMOLI SPECIES, NMOLI= 1 1 D2 4 2 2 0 0 1 0 3 1 115 113 0 00000 -1.5400E 01 0.0000E 00 0.0000E 00 0.0000E 2 115 121 000 00000 -1.0500E 01 0.0000E 00 3.0000E 00 3.0000E 3 115 111 *14 00000 -2.5000E 01 0.0000E 00 5.0000E 00 5.0000E * TEST ION SPECIES CARDS: NIONI ION SPECIES, 1 1 D2+ 4 2 2 1 0 1 0 3 -1 4 115 111 *14 00000 -1.0500E 01 0.0000E 00 4.3000E 00 4.3000E 5 115 00000 *24 -1.5500E 01 0.0000E 00 0.2500E 00 0.2500E 6 115 121 000 30300 7.0000E 00 0.0000E 00 7.0000E 00 0.0000E 00 00 00 NIONI= 00 00 00 As above, ∗ stands for the species index of the D+ bulk ion. Note that for reaction 6, the energy dependence of the cross section leads to a mean electron energy loss per event E el of about 0.88 · Te , see reference [33]. This approximation is used in the default model for the electron energy loss Eel per collision and for the total kinetic energy release EK shared by the two product atoms. The reaction no. 7 above: AMJUEL H.8 2.1.14, provides values very close to this, by an independent fit to the correct electron energy loss for this process ( ) d ln⟨σve ⟩ ⟨σve Eel ⟩ = Eel · ⟨σve ⟩ = kTe ⟨σve ⟩ · 3/2 + (4.4) d ln(kTe ) The velocity distribution of the dissociation products is isotropic in the center of mass system (taken to be the system moving with the incident molecule), and the dissociation energy is shared by the dissociation products so as to preserve momentum. I.e., in case of unsymmetrical hydrogenic molecules the two dissociation products to not receive the same share of the dissociation kinetic energy release, but instead this is distributed inversely proportional to their masses. In case of mixed molecules, such as DT , HT or HD some rate coefficients are automatically split into two half and assigned to the proper product atomic particles. For hydrogenic bulk ions (H + , D+ , T + ) default volume recombination rates are available [34]. Further examples to be written 2.4.2 Neutral-Neutral collisions in BGK approximation In the example considered here we have the 4 neutral-neutral collision processes: D on D2, D on D and D2 on D2, D2 on D. In input block 4 there are three BGK relaxation rate coefficients (note: the rate coefficient for D2 on D is equal to the one for D on D2, hence 3 rate coefficients are sufficient). 126