Download LAB_LEDI_II _UM
Transcript
LABORATORY
ELECTRONIC DATA INTERCHANGE
PHASE II (LEDI II)
USER MANUAL
LA*5.2*46/LR*5.2*222
Version 5.2
February 2002
Department of Veterans Affairs
Veterans Health Administration
Office of Information
Preface
The Veterans Health Information Systems and Architecture (VISTA) Laboratory
Electronic Data Interchange (LEDI II) User Manual Version 5.2 for patch LA*5.2*46
and patch LR*5.2*222 provides the Department of Veterans Affairs Medical Center
(DVAMC) Information Resource Management (IRM) staff, Laboratory Information
Managers (LIMs), and other DVAMC users with a straightforward means for
implementing and utilizing the LEDI II software.
The LEDI II User Manual (Patch LA*5.2*46 and LR*5.2*222) focuses on easy-tofollow step-by-step instructions. This user manual consists of the following sections:
Introduction: This section conveys the major functions, purposes, and how the
software accomplishes the objectives.
Orientation: This section defines the symbols and computer screen dialogue used
throughout the LEDI II User Manual.
Package Management: This section contains mail groups, alerts, security keys,
special VA FileMan access codes, and other security measures information included
with the LEDI II software release.
Package Operation: This section describes what the user needs to know in order to
competently operate the LEDI software application and where to access the on-line
documentation.
LEDI II Implementation Instructions: This section contains detailed setup
instructions to successfully implement the LEDI II software application. LEDI II
setup instructions must be performed in sequence, by IRM and LIM staff. The setup
instructions are listed in step-by-step order.
LEDI II Setup Checklist: The checklist is provided to ensure that the LEDI II
setup implementation process has been completed as instructed in the package
operation section of this user manual.
Glossary: This section contains the glossary of terms that are related to the LEDI
software application.
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
iii
Preface
iv
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
Table of Contents
PREFACE---------------------------------------------------------------------------------------------------------III
INTRODUCTION ------------------------------------------------------------------------------------------------9
OVERVIEW --------------------------------------------------------------------------------------- 9
VISTA Laboratory Electronic Data Interchange (LEDI) -------------------------------------------9
VISTA Laboratory Electronic Data Interchange Phase II (LEDI II) -----------------------------9
New Functionality----------------------------------------------------------------------------------------- 10
Patient Lookup--------------------------------------------------------------------------------------------- 10
LEDI II ENHANCEMENTS AND MODIFICATIONS ------------------------------------------------ 11
Enhancements: --------------------------------------------------------------------------------------------- 11
1. Sending/Receiving Laboratory HL7 Messages---------------------------------------- 11
2. TCP/IP Protocol as a Communication Protocol--------------------------------------- 11
3. Building Lab Test Orders------------------------------------------------------------- 11
4. Processing NTE Segments ----------------------------------------------------------- 11
5. Storing Test Reference Ranges ------------------------------------------------------- 12
6. Specifying Final or Incomplete Lab Test Results -------------------------------------- 12
7. New SM40 Shipping Codes --------------------------------------------------------- 12
8. Building a Shipping Manifest---------------------------------------------------------- 12
9. Closing a Shipping Manifest ---------------------------------------------------------- 12
10. Result (ORU) Messages --------------------------------------------------------------- 13
11. Result (ORU) Messages --------------------------------------------------------------- 13
12. Referral Patient Multi-purpose Accession [LRLEDI] option---------------------------- 13
Modification ------------------------------------------------------------------------------------------------ 14
VA FileMan Database Server Call--------------------------------------------------------- 14
VISTA BLOOD BANK SOFTWARE V5.2 DEVICE PRODUCT LABELING STATEMENT14
FLOW CHART ---------------------------------------------------------------------------------- 15
ORIENTATION------------------------------------------------------------------------------------------------- 17
COMPUTER DIALOGUE-------------------------------------------------------------------------- 17
USER ENTRY RESPONSE ------------------------------------------------------------------------- 17
RETURN SYMBOL ------------------------------------------------------------------------------- 17
TAB SYMBOL ----------------------------------------------------------------------------------- 17
BRACKET SYMBOL------------------------------------------------------------------------------ 17
REFERENCES ------------------------------------------------------------------------------------ 18
PACKAGE MANAGEMENT ------------------------------------------------------------------------------- 19
CONTINGENCY PLANNING----------------------------------------------------------------------- 19
FILE SECURITY---------------------------------------------------------------------------------- 19
SECURITY KEYS--------------------------------------------------------------------------------- 19
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
5
Introduction
ELECTRONIC SIGNATURE------------------------------------------------------------------------ 20
PURGING CAPABILITIES ------------------------------------------------------------------------- 20
INSTRUMENT INTERFACING --------------------------------------------------------------------- 20
REMOTE SYSTEMS ------------------------------------------------------------------------------ 20
LEDI II MAIL GROUPS ------------------------------------------------------------------------- 22
LAB MESSAGING Mail Group:------------------------------------------------------------------------ 22
LA7V* Mail Groups:-------------------------------------------------------------------------------------- 22
ALERTS ----------------------------------------------------------------------------------------- 23
LEDI II IMPLEMENTATION INSTRUCTIONS----------------------------------------------------- 25
LEDI II TWO-PART IMPLEMENTATION SETUP-------------------------------------------------- 25
IRM and LIM Setup Instructions------------------------------------------------------------------------ 26
Step #1 - IRM: INSTITUTION file (#4) Setup -------------------------------------------- 26
Step #2 IRM: Create Local LEDI Mail Groups -------------------------------------------- 28
Step #3 IRM: Schedule the Lab Messaging Nightly Cleanup [LA7TASK NIGHTY] option29
Example: Scheduling the Lab Messaging Nightly Cleanup [LA7TASK NIGHTY] option to
run daily.-------------------------------------------------------------------------------- 29
Step #4 LIM: Collection and Host Facilities Information Requirements --------------------- 30
Host Facility ---------------------------------------------------------------------------- 30
Collection Facility----------------------------------------------------------------------- 31
Step #4 continu for COLLECTION facility------------------------------------------------ 32
Step #5 LIM (collection facility): Lab Shipping Files Setup --------------------------------- 33
Collection Facility: ---------------------------------------------------------------------- 33
Step #5 continue for COLLECTION facility----------------------------------------------- 34
Collection Facility: ---------------------------------------------------------------------- 34
Example: CME Edit Shipping Method [LA7S EDIT 62.92] option--------------------- 34
Collection Facility: ---------------------------------------------------------------------- 35
Example: CDE Edit Shipping Condition [LA7S EDIT 62.93] option------------------- 35
Collection Facility: ---------------------------------------------------------------------- 36
Example: CTE Edit Shipping Container [LA7S EDIT 62.91] option------------------- 36
Step #5 continue for COLLECTION facility----------------------------------------------- 37
Collection Facility: ---------------------------------------------------------------------- 37
Commercial Reference Laboratory (i.e., non-VA facility) Setup Example ------------- 37
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option----------------- 37
Step #5 continues for COLLECTION facility---------------------------------------------- 41
Step #5 continues for COLLECTION facility---------------------------------------------- 45
Step #6 LIM (host facility): LAB SHIPPING CONFIGURATION file (#62.9) Setup ----- 56
Host Facility:---------------------------------------------------------------------------- 56
Commercial Reference Laboratory (non-VA facility) Setup --------------------------- 56
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option ---------------- 56
Step #6 continues for HOST facility-------------------------------------------------------- 57
Step #6 continued for HOST facility ------------------------------------------------------- 59
Step #6 continues for HOST facility-------------------------------------------------------- 62
6
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
Introduction
VA Facility Setup-------------------------------------------------------------------------- 62
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option----------------- 62
Step #7 LIM: LEDI II Setup for Laboratory Auto-Instruments and HL7 Environment ------ 65
Step #7 continues - LEDI II Setup for Laboratory Auto-Instruments and HL7 Environment 66
Example: LSU LEDI Setup ----------------------------------------------------------- 66
Step #8 IRM: Completing the HL7 Environment Setup------------------------------------- 73
1a. TCP/IP Interfaces------------------------------------------------------------------- 73
IRM: Complete the HL7 Environment Setup. ----------------------------------------- 73
1b. MailMan Interfaces ----------------------------------------------------------------- 75
LEDI II CONFIGURATION SETUP CHECKLIST--------------------------------------------------- 77
CHECKLIST FOR COLLECTION FACILITY SETTING UP HOST FACILITY ---------------------------- 77
CHECKLIST FOR HOST FACILITY SETTING UP COLLECTION FACILITY ---------------------------- 79
PACKAGE OPERATION ------------------------------------------------------------------------------------ 81
LEDI II DOCUMENTATION RETRIEVAL FORMATS ---------------------------------------------- 81
File Names
Contents
Retrieval Formats----------------- 81
LEDI II DOCUMENTATION RETRIEVAL LOCATIONS --------------------------------------------- 81
Website Locations: ---------------------------------------------------------------------------------------- 82
Laboratory Version 5.2 Home Page:------------------------------------------------------- 82
VISTA Documentation Library (VDL):----------------------------------------------------- 82
LEDI II NEW AND MODIFIED OPTIONS --------------------------------------------------------- 83
Edit Required Test Information [LA7S MANIFEST TEST REQ INFO] option------------ 83
Retransmit LEDI Lab Results [LA7S RESULTS RETRANSMIT] option: ----------------- 83
Print LEDI Pending Orders [LA7S PENDING PRINT LEDI] option: --------------------- 83
Display Lab Universal Interface Message [LA7 PRINT LAB UI MESSAGE] option:------ 83
LEDI II MENUS AND OPTIONS------------------------------------------------------------------ 84
LEDI MENU AND OPTION EXAMPLES ---------------------------------------------------------- 91
Electronic Catalog Information Entry [LA7S CATALOG ENTRY] ---------------------------- 91
Example: ENT Electronic Catalog Information Entry option -------------------------------- 91
View Individual Electronic Catalog Entry [LA7S VIEW INDIVIDUAL ENTRY] ------------ 92
Example: IND View Individual Electronic Catalog Entry option----------------------------- 92
Referral Patient Multi-purpose Accession [LRLEDI] ---------------------------------------------- 93
Example 1: Manual Accession ------------------------------------------------------------- 93
Example 2: Using Bar Code Reader ------------------------------------------------------- 95
Example 2: Using Bar Code Reader (continues)-------------------------------------------- 96
Turnaround times By Urgency [LR TAT URGENCY] --------------------------------------------- 97
Example: Turnaround times by Urgency---------------------------------------------------- 97
Example: Turnaround times by Urgency (continues) ---------------------------------------- 98
GLOSSARY------------------------------------------------------------------------------------------------------ 99
CAP:-------------------------------------------------------------------------------------- 99
Collection facility:-------------------------------------------------------------------------- 99
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
7
Introduction
Data Fields:-------------------------------------------------------------------------------- 99
Data Type (DT):--------------------------------------------------------------------------- 99
Electronic Catalog:------------------------------------------------------------------------- 99
Element Name: ---------------------------------------------------------------------------- 99
Host facility: ------------------------------------------------------------------------------- 99
LEDI: ----------------------------------------------------------------------------------- 100
Length (LEN):--------------------------------------------------------------------------- 100
NLT: ------------------------------------------------------------------------------------ 100
Optionally (R/O/C):---------------------------------------------------------------------- 100
PID:------------------------------------------------------------------------------------- 100
Repetition (RP/#): ----------------------------------------------------------------------- 100
Segments: ------------------------------------------------------------------------------- 100
Shipping Manifest:----------------------------------------------------------------------- 100
Sequence Number (SEQ):--------------------------------------------------------------- 100
Table (TBL#):--------------------------------------------------------------------------- 101
TCP/IP: --------------------------------------------------------------------------------- 101
Trading Partners: ------------------------------------------------------------------------ 101
UI: -------------------------------------------------------------------------------------- 101
UID:------------------------------------------------------------------------------------- 101
VAMC: --------------------------------------------------------------------------------- 101
VISN:----------------------------------------------------------------------------------- 101
VISTA ---------------------------------------------------------------------------------- 101
VISTA Laboratory UI and LEDI HL7 Interface Standard Specifications V. 1.1: ---------- 101
Workload System:----------------------------------------------------------------------- 101
8
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
Introduction
Overview
The Veteran Integrated Service Network (VISN) mission is to consolidate electronic
lab test ordering and lab test result reporting throughout all Veterans Affairs (VA)
Medical Care Facilities Laboratories within a VISN, between VISNs, and non-VA
organizations (i.e., commercial reference laboratories). The existing VISTA
Laboratory Electronic Data Interchange (LEDI) software application required
modifications and enhancements to accomplish this mission without diminishing the
quality of services in patient medical care.
VISTA Laboratory Electronic Data Interchange (LEDI)
The VISTA Laboratory Electronic Data Interchange (LEDI) software provided the
Laboratory V. 5.2 software package and Laboratory’s Automated Instrument (AI)
software application the following features and functionalities:
•
•
•
•
•
Laboratory Electronic Data Interchange (LEDI) – Electronic Messaging
Electronic Lab Test Ordering
Electronic Lab Test Results Reporting
Bar-code Specimen Accessioning
Workload
VISTA Laboratory Electronic Data Interchange Phase II (LEDI II)
The VISTA Laboratory Electronic Data Interchange Phase II (LEDI II) software
application provides electronic messaging for Lab Test Ordering and Lab Test
Results Reporting between VA Health Care Facilities laboratories based on the
Health Level Seven (HL7) Version 2.3 Standard Specification and VISTA Health
Level Seven (HL7) Version 1.6 Standard Specification. These Specifications are used
as the basis for defining VISTA Laboratory Universal Interface (UI) and LEDI HL7
Interface Standard Specification Version 1.2.
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
9
Introduction
New Functionality
LEDI II software application provides the following new features and functionality
for Laboratory V. 5.2 Package and the Automated Instrument (AI) software
applications.
•
Laboratory Electronic Data Interchange (LEDI) - Electronic Messaging
•
Electronic Lab Test Ordering
•
Bar-code Specimen Accessioning
•
Electronic Lab Test Results Reporting
•
Workload
Patient Lookup
The patient lookup functionality relieves accessioning personnel from manually
entering patient’s demographics. The lookup functionality checks the PATIENT file
(#2), REFERRAL PATIENT file (#67), and LAB PENDING ORDERS ENTRY file
(#69.6) when using bar code scanning. This patient lookup functionality will reduce
or eliminate typographical mistakes and decreases the possibility of duplicate
entries. The Patient demographics displays the following fields:
⇒ Full Name
⇒ Date of birth (DOB)
⇒ Patient Identification (SSN)
⇒ Sex
10
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
Introduction
LEDI II Enhancements and Modifications
Enhancements:
1. Sending/Receiving Laboratory HL7 Messages
New functionality has been added for sending and receiving Laboratory HL7
orders/results messages with non-VA facilities (Commercial Reference Laboratories).
2. TCP/IP Protocol as a Communication Protocol
Additional new support is added for TCP/IP protocol as a communication protocol for
the transmission of LEDI HL7 messages (i.e., requiring patch HL*1.6*19 to have
been successfully installed). The implementation of this communication protocol in
lieu of VA MailMan protocol will significantly increase transmission speed of
messages and reduce system overhead/storage. Both client and server must
coordinate the switch to this new communication protocol transmission method. The
following two logical links are established to exchange messages:
•
•
Server (listener) logical link used to receive and process messages.
Client logical link used to transmit messages.
3. Building Lab Test Orders
The process of building orders in the LAB PENDING ORDERS ENTRY file (#69.6)
has been enhanced to use information from the LAB SHIPPING
CONFIGURATION file (#62.9), to create orders based on the host site’s specified
collection sample and urgency.
4. Processing NTE Segments
The capability to process NTE segments following OBX segments have been
enhanced to site-selectable. Comments found attached to specific lab test results are
stored as comments. The AUTO INSTRUMENT file (#62.4), CHEM TESTS subfile
(#30), REMARK PREFIX field (#19) can be used to specify a comment prefix that is
made a part of the comment.
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
11
Introduction
5. Storing Test Reference Ranges
Storing test reference ranges, abnormal flags, and the name of the performing
laboratory have been enhanced to site-selectable. These parameters are stored as
COMMENTS with the results. This information is prefixed with the REMARK
PREFIX field (#19), of the AUTO INSTRUMENT file (#62.4).
6. Specifying Final or Incomplete Lab Test Results
The ability to specify whether final or incomplete lab test results are processed has
been enhanced to site-selectable.
7. New SM40 Shipping Codes
The new SM40 shipping code (required shipping information update) has been
added to the LAB ELECTRONIC CODES file (#64.061). The SM40 shipping code is
used for recording this information in the LAB SHIPPING EVENT file (#62.85).
8. Building a Shipping Manifest
The process for building a shipping manifest has been enhanced to allow the user to
specify the starting and ending accession numbers to check within an accession area.
This feature will shorten the manifest building time when searching a yearly
accession area with a large number of accessions. This new feature allows the site to
skip the accessions numbers that were completed earlier in the year and no longer
need to be checked to determine if any of the tests meet the criteria for building to a
manifest.
9. Closing a Shipping Manifest
The process of closing a Shipping Manifest has been enhanced to perform new
checks for the following required information:
•
•
•
12
Each lab test has an order NLT code assigned.
When shipping to a facility that does not use VA NLT codes for lab test
ordering, the appropriate non-VA lab test codes/names is designated for all
tests on the Shipping Manifest.
Site-specified required patient/specimen information is entered.
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
Introduction
10. Result (ORU) Messages
LEDI II Result (ORU) Messages includes the test interpretation found in
LABORATORY TEST file (#60) as a comment (i.e., NTE segment) following the lab
test results. Lab test results storage is checked against the VA FileMan input
transform for the data name used to store the Lab test results in LAB DATA file
(#63). If the lab test results does not pass the VA FileMan input transform check,
then an error message is generated via the Lab Universal Interface error reporting
process. Result values “canc”, “pending”, or “comment” are stored in LAB DATA file
(#63).
11. Result (ORU) Messages
The process that triggers LEDI II Result (ORU) Message generation, during lab
result verification, has additional checks to confirm that the results being returned to
the collecting site match the ordering information from the collecting facility. Patient
and specimen identifiers used by the host and collection facilities are checked for
expected consistency. When identifiers do not match, the HL7 message transmission
will be aborted and an error message generated via the Lab Universal Interface error
reporting process. Error messages #40-45 have been added to the LA7 MESSAGE
LOG BULLETINS file (#62.485) to support this functionality. These are additional
safety checks being incorporated into the software in response to a recent patient
safety alert involving the Laboratory V. 5.2 Package.
12. Referral Patient Multi-purpose Accession [LRLEDI] option
LEDI II software contains additional support when using the Referral Patient Multipurpose Accession [LRLEDI] option to accession specimens thru the Laboratory
Electronic Data Interface (LEDI). LEDI II now builds the LAB PENDING ORDERS
ENTRY file (#69.6) with additional information concerning mapping to local host test
urgency. This local urgency information, stored in File (#69.6), is utilized when
accessioning LEDI specimens at the host laboratory.
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
13
Introduction
Modification
VA FileMan Database Server Call
Laboratory Package V. 5.2 previously used a classical VA FileMan DIE Call when
storing comments in LAB DATA file (#63). This VA FileMan DIE Call did not allow
the comment to contain a semi-colon “;” character because FileMan used this
character to parse fields requiring editing in the DR string. This function has been
changed to the VA FileMan Database Server Call, which does not have this
restriction. Comments can now contain the semi-colon “;”character.
VISTA BLOOD BANK SOFTWARE V5.2 DEVICE PRODUCT
LABELING STATEMENT
12. June 10, 1999
VISTA Laboratory Package patches LA*5.2*46/LR*5.2*222 contain no changes to
software controlled by VHA DIRECTIVE 99-053, titled VISTA BLOOD BANK
SOFTWARE.
14
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
Introduction
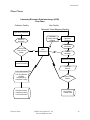
Flow Chart
Laboratory Electronic Data Interchange (LEDI)
Flow Chart
Host Facility
Collection Facility
Specimen Tubes/Shipping Manifest
Specimen Collection
Collection
Facility Orders
Pending
Specimen
Accession
Host Facility
Specimen
Receiving
Specimen
Accession
Send out Lab
Tests To Host
Facility
Host
HL7
Messaging
Status
Message
Specimen Analysis
Collection Facility
Normal Processing
Lab Test
Results
Auto-Instrument File
Lab Test Results
Pending
Acceptance by
Collection Facility
Collection Facility
Patient
Collection Facility
Patients Verified
Lab Test Results
Host Facility
Patient Data
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
15
Introduction
16
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
Orientation
This section defines the symbols and computer screen dialogue used throughout the
LEDI II User Manual.
Computer Dialogue
The computer dialogue appears in Courier font, no larger than 10 points.
Example: Courier font 10 points
User Entry Response
User entry response appears in boldface type Courier font, no larger than 10 points.
Example: Boldface type
Return Symbol
User response to computer dialogue is followed by the <RET> symbol, which appears
in Courier font, no larger than 10 points, and bolded.
Example: <RET>
Tab Symbol
User response to computer dialogue is followed by the <Tab> symbol, which appears
in Courier font, no larger than 10 points bold.
Example: <Tab>
Bracket Symbol
The bracket symbols [ ] are used for notations within the computer screen dialogue.
Example: [ ]
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
17
Orientation
References
The following references may be helpful when installing and implementing the LEDI
II software application:
•
Kernel Systems Manual V. 8.0
•
Kernel Toolkit V. 7.3
•
Laboratory Universal Interface Patch LA*5.2*17-Patch LR*5.2*65 V. 1.0
•
VA FileMan V. 21.0
•
VA MailMan V. 7.1
•
VISTA Health Level Seven Site Manager & Developer Manual Version 1.6*56
•
VISTA Laboratory Health Level Seven (HL7) Interface Standard
Specifications V. 1.2
•
Health Level Seven Standard Version 2.3.1 © 1997 by the Health Level
Seven, Inc.
NOTE: Copies of the HL7 standards are available via the HL7 (VISTA Messaging V.
1.6 Home Page at http://vista.med.va.gov/hl7/
18
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
Package Management
The LEDI II software application consists of the following mail groups, alerts,
security keys, VA FileMan access codes, and security information.
Contingency Planning
Each facility using the LEDI IIsoftware application must develop a local
contingency plan to be used in the event of application problems in a live
environment. The facility contingency plan must identify procedures used for
maintaining the functionality provided by the software in the event of a system
outage.
File Security
LEDI II changes and enhancements do not modify any existing file security
schemes. New files exported by the patch installation have no file security applied.
However, VA FileMan security access Ll code is recommended if file security is
deemed necessary by the facilities.
Security Keys
LRDATA key
The LRDATA security key is used to identify personnel that are allowed to edit or
change test results that were done by another laboratory. If an associated division
did not generate the results, this security key is required to change the results. The
Laboratory responsible for the original lab test results must send and edit the LEDI
II messages to correct errors.
LRLEDI Security Key
The LRLEDI security key is used to identify users that are charged with the setup
and configuration of the LEDI II software functionality. In most instances, the
LRLIASON key holder would also hold this key.
NOTE: LEDI II setup options are exported with the LRLEDI security key already
assigned.
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
19
Package Management
Electronic Signature
There are no electronic signatures utilized in the VISTA LEDI II software
application.
Purging Capabilities
LEDI II purging capabilities are provided through the Universal Interface (UI)
software. The Lab Messaging Nightly Cleanup [LA7TASK NIGHTY] option is used
to purge messages in the LA7 MESSAGE QUEUE file (#62.49) when the messages
become eligible for purging. This option must be scheduled to run daily, preferably
during a period when activity in the Lab Messaging (i.e., UI and LEDI software) is at
a minimum. See the Lab Messaging Nightly Cleanup [LA7TASK NIGHTY] option
description for additional information and functionality. Purging of messages for
each configuration can be set in the LA7 MESSAGE PARAMETER file (#62.48)
using the Grace Period For Messages field (#3). The Default value is 3 days.
Instrument Interfacing
LEDI II is an enhancement to Laboratory V. 5.2 and does not specifically involve
instrument interfacing. All electronic communication is via established HL7
procedures for transmitting messages. The VISTA Laboratory HL7 Interface
Standards have been enhanced to accommodate expanded messaging capability.
Remote Systems
Remote systems are defined as any computer system sending to and receiving HL7
messages from the Host laboratory computer system. In VA-to-VA configurations –
this can be within the same system (i.e. multidivisional sites), or it can be from one
system to another system, (i.e. non-related sites). In the case of non-VA computer
systems, connection to remote systems is done via Generic Instrument Manager
(GIM). The GIM is the remote system, not the non-VA computer system.
20
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
Package Management
NOTE: As of the release date of this software, there is no national solution to
implement a laboratory electronic HL7 standard interface to a facility or other entity
outside the VA wide area network. National communication security concerns are
still being debated by the Department of Veterans Affairs (VA) and are awaiting
resolution. At present, a VA medical center, which intends to utilize this software in
conjunction with an interface to a commercial reference laboratory or other non-VA
information system entity, will need to coordinate with the vendor an acceptable
communication method. Any implementation will need to be approved by the facility
and VISN Information Security Officer (ISO) and meet current VA security
requirements for external electronic connections. See VHA Directive 6212, Security
of External Electronic Connections and VHA Directive 6210, Automated Information
Systems (AIS) Security for additional information and guidance. Additional security
information may be obtained from the Health Information Security Service (HISS)
web page at http://vaww.va.gov/miss/
Implementation of a HL7 messaging interface between the VA VISTA Laboratory
package and a non-VA information system consists basically of three parts:
•
•
•
VISTA Laboratory LEDI II software.
Certified communication software and hardware.
Non-VA information system capable of sending and receiving Laboratory HL7
order and result messages.
All three parts must be functional to utilize the capabilities of this LEDI II software
patch. The implementation, setup, and configuration of vendor provided hardware
and software are NOT addressed by this documentation. Consult the vendor provided
documentation and instructions to interface to the VISTA Laboratory package.
LEDI II utilize the HL7 V. 2.3.1 Standard and the VISTA HL V. 1.6 software to
transmit and receive HL7 messages via VA MailMan or Transmission Control
Protocol/Internet Protocol (TCP/IP). The software transmits unsolicited order and
result messages and receives acknowledgments back confirming or opposing the
proper delivery of the messages. One order message is transmitted for every manifest
or lab delivery to a remote facility system. One result message is sent to each facility
per verifying session. To reduce the turn around time and network mail traffic,
TCP/IP is recommended as the message delivery medium.
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
21
Package Management
LEDI II Mail Groups
The following mail groups are required to successfully utilize the LEDI II
functionality. LEDI II uses VA MailMan or TCP/IP as the medium to transmit and
receive data from remote systems.
LAB MESSAGING Mail Group:
This is a general mail group used by the LAB Universal Interface and LEDI II
software to address alerts when conditions are detected requiring review and/or
corrective action. The members of this mail group should, at the minimum, include
the LIM and selected Lab and IRM personnel responsible for maintenance and
support of the LAB Universal Interface and LEDI IIsoftware. The software utilizes
several Kernel Alerts. These alerts are triggered and sent to members of the LAB
MESSAGING mail group for the following conditions:
•
When the scheduled tasked Lab Messaging Nightly Cleanup [LA7TASK
NIGHTY] option or the Start/Stop Auto Download Background Job [LA7 ADL
START/STOP] option is run and more than 500 entries are found scheduled for
downloading via the Lab Universal Interface an alert is sent notifying members
of this condition.
•
When the scheduled tasked Lab Messaging Nightly Cleanup [LA7TASK
NIGHTY] option or the Lab Messaging File Integrity Checker [LA7 CHECK
FILES] option is run and LA7 MESSAGE QUEUE file (#62.49) is found to have
bad entries and/or cross-references. An alert is sent notifying members of this
condition.
LA7V* Mail Groups:
These mail groups are used by the HEALTH LEVEL SEVEN package for
transmitting LEDI HL7 messages when VA MailMan is selected as the HL7
communication protocol. These mail groups are created by the LEDI Setup [LA7V
SETUP] option using the LA7V name space concatenated with the receiving facility’s
primary station number. For example, to send HL7 messages to Dallas OI Field
Office, VA Station Number 170, the LEDI Setup option would create a mail group
LA7V 170. Other than the remote member added by the LEDI Setup option, no local
or remote members should be added to these mail groups.
22
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
Package Management
Local Mail Groups:
It is highly recommended that each facility designate local mail groups for receiving
the “New Results”, “Orders Received”, and the “Error on Message “ alerts associated
with the LEDI II software application. Please review the following section on
ALERTS for information on the various alerts that are generated by LEDI II.
Alerts
LEDI II software uses the LA7 MESSAGE PARAMETER file (#62.48) for sending
alerts to the mail group specified in the Alert Condition subfile (#68.481). The facility
should designate a local mail group to use for notification of the alerts. These alerts
and the associated mail group are configured using the LEDI Setup [LEDI SETUP]
option, within the message configuration section of this option. The following are the
three types of alerts that can be turned on for each configuration:
1. New Results Alert - notifies members when the Lab Universal Interface software
has processed an HL7 message containing test results. An example of this
information type of alert is: “Lab Messaging – New results received for LA7V HOST
578”.
2. Error Alert - notifies members of error conditions encountered during the
processing of a Laboratory Universal Interface message. Recommend that sites
utilize the LAB MESSAGING mail group to notify local users of error conditions
within the Laboratory Universal Interface/LEDI software. Processing the alert allows
the user to view/print the error message and the associated HL7 message. An
example of this action type of alert is: “Lab Messaging error #17 on message
#246164473”.
3. New Orders Received Alert – notifies members when the Laboratory Universal
Interface software has received electronic orders related to a collecting facility’s
shipping manifest. An example of this information type of alert is: “Lab Messaging Manifest# 537-20011030-3 received from LA7V COLLECTION 537”.
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
23
Package Management
24
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
This section contains detailed instructions to successfully implement the LEDI II
software application. LEDI II setup instructions must be performed in sequence, by
IRM and LIM staff. The setup instructions are listed in step-by-step order.
NOTE: A checklist is provided at the end of this implementation section to ensure
that the LEDI II implementation process has been completed as instructed in this
section.
LEDI II Two-Part Implementation Setup
The LEDI II software application requires the following two-part implementation
setup:
Collection facility laboratories must be setup to perform the following:
§
Create an electronic Shipping Manifest (i.e., specifying lab tests to be
performed by the receiving host facility laboratory).
§
Transmit the Shipping Manifest to the host facility laboratory.
§
Transport the specimens to the host facility laboratory.
Host facility laboratories must be setup to perform the following:
§
Receive the electronic Shipping Manifest List (transmitted by the collection
facility laboratory).
§
Scan the Shipping Manifest upon specimen arrival at the host facility.
§
Bar codes the specimen arrival at the host facility.
§
Perform the specified lab tests and verifies the lab test results.
§
Transmit lab test results back to the collection facility.
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
25
LEDI II Implementation Instructions
IRM and LIM Setup Instructions
NOTES:
The collection and host facilities IRM and LIM staff must coordinate the LEDI II
implementation setup once the LA*5.2*46 and LR*5.2*222 patches have been
installed.
The IRM and LIM staff must perform the LEDI II implementation setup
instructions in the sequence specified below.
Step #1 - IRM: INSTITUTION file (#4) Setup
LEDI II software application uses the INSTITUTION file (#4), NAME field (#.01),
DOMAIN field (#60), AGENCY CODE field (#95), and the STATION NUMBER field
(#99), to identify both the collection and host facilities.
IRM: Use VA FileMan [DIUSER], Enter or Edit File Entries [DIEDIT] option to
appropriately define the following four fields:
NAME field (#.01): Enter the name of the instiution for both the host and collection
facilities. Applies ONLY for non-VA entries. The Institution Master File maintains
nationally controlled entries.
DOMAIN field (#60) (VA facilities ONLY): If this field is null the VA facilites must
manually add the server to the mail group that is created by the LEDI Setup [LA7V
SETUP] option. Using VA MailMan as the HL7 transport protocol is ONLY
supported when both collection and host facilities are VA faciltities. Other non- VA
facilities using VA MailMan for their HL7 message transfer must update the
DOMAIN field (#60) for the associated facilities. Updating this field allows the LEDI
Setup [LA7V SETUP] option to automatically setup the HL7 remote server for the
host or collection facility laboratories.
26
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
NOTES:
The LEDI Setup [LA7V SETUP] option also creates an LA7V xxx mail group, (i.e.,
xxx is the VA facility’s station number). This mail group uses the LA7V namespace
concatenated with the receiving facilities primary station number.
If the DOMAIN field (#60) contains data, the LEDI Setup [LA7V SETUP] option
automatically adds the HL7 remote server to the MAIL GROUP file (#3.8), REMOTE
MEMBERS field (#12).
Faciltities using TCP/IP for their HL7 transport protocol will require additional
setup of the HL7 links by IRM after the LEDI Setup [LA7V SETUP] option is used to
create the necessary HL7 links.
AGENCY CODE field (#95): This field indicates to the LEDI II software whether
the facility is a VA or non-VA facility. Applicable only for non-VA entries. The
Institution Master File maintains nationally controlled entries. The AGENCY
CODE field (#95) should be defined as follows:
•
VA Facilities: VA
•
Military Facilities (by the appropriate branch of service): AIR FORCE,
ARMY, or NAVY)
•
Indian Health Service: HIS
•
Commercial reference laboratories and non-US government health care
facilities: OTHER
NOTE: The LEDI Setup [LA7V SETUP] option uses the AGENCY CODE field (#95)
to determine the status of the facility and how to setup the LEDI II software
application.
STATION NUMBER field (#99) (VA facilities ONLY, unique number): The
Institution Master File maintains nationally controlled entries. LEDI II uses the VA
facility station number, for configuring VA facilities and creating the Lab Shipping
files setup entries using the Lab Shipping Management [LA7S MGR MENU] menu.
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
27
LEDI II Implementation Instructions
Step #2 IRM: Create Local LEDI Mail Groups
IRM: Use VA FileMan [DIUSER], Enter or Edit File Entries [DIEDIT] option to
create local LEDI mail groups and to add designate members. It is highly
recommended that the LIM be designated as a member of all the LEDI mail groups.
The LEDI mail groups are also used by the LEDI alert system.
NOTES:
DO NOT add any local or remote members to the LAV7* mail groups.
DO NOT use the LAV7*mail groups for local LEDI alerts.
28
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
Step #3 IRM: Schedule the Lab Messaging Nightly Cleanup [LA7TASK
NIGHTY] option
IRM: Use Taskman Management [XUTM MGR], Schedule/Unschedule [XUTM
SCHEDULE] option to schedule the Lab Messaging Nightly Cleanup [LA7TASK
NIGHTY] option to run daily. This scheduled option will purge all data in the LA7
MESSAGE QUEUE file (#62.49).
Example: Scheduling the Lab Messaging Nightly Cleanup [LA7TASK NIGHTY]
option to run daily.
Select OPTION to schedule or reschedule: LA7TASK NIGHTY<RET>
Nightly Cleanup
...OK? Yes//<RET>
(Yes)
Edit Option Schedule
Option Name: LA7TASK NIGHTY<RET>
Menu Text: Lab Messaging Nightly Cleanup
Lab Messaging
TASK ID: 8278506
QUEUED TO RUN AT WHAT TIME: JUL 14,1997@23:15<RET>
DEVICE FOR QUEUED JOB OUTPUT: NULL DEVICE;P-OTHER;132;64<RET>
QUEUED TO RUN ON VOLUME SET:<RET>
RESCHEDULING FREQUENCY: 1D<RET>
TASK PARAMETERS:<RET>
SPECIAL QUEUEING:<RET>
______________________________________________________________________________
Exit
Save
Next Page
Refresh
Enter a command or '^' followed by a caption to jump to a specific field.
COMMAND: N
Press <PF1>H for help
Insert
Edit Option Schedule
Option Name: LA7TASK NIGHTY<RET>
_____________________________________________________________________
VARIABLE NAME: LA7FIX
VARIABLE NAME: LA7ION
VARIABLE NAME: LA7LOG
VARIABLE NAME:
VARIABLE NAME:
February 2002
VALUE: 0
VALUE: "IRM DEVELOP LASER1"
VALUE: 1
VALUE:
VALUE:
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
29
LEDI II Implementation Instructions
Step #4 LIM: Collection and Host Facilities Information Requirements
Host Facility
The host facility laboratory must provide the collection facility laboratory with the
following information requirements:
•
Specimen collection requirements
•
Schedule of testing
•
List of tests for panel tests
•
Normal range and critical values
•
Patient prep instructions load
•
Provide a list of National Lab Test (NLT) codes used by the host facility
laboratory. (VA Host facility only)
NOTE: The Electronic Catalog Menu [LAS7 CATALOG MENU] can be used to
produce a list of the host facility NLT codes. This menu is located on the Lab
Shipping Management Menu [LA7S MGR MENU], which is located on the Lab
Liaison Menu [LRLIAISON].
30
•
NLT order code to be used to order the test
•
NLT Result/LOINC codes used to report the results
•
Accession Area numeric identifier (VA Host facility only)
•
Bar-code symbology and label stock
•
Provide the collection facility laboratory with a list of lab tests that are
available for processing. Note: The Electronic Catalog Menu [LAS7
CATALOG MENU] may also be used to produce a list of the host facility
available lab tests requiring processing.
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
Step #4 continued for HOST facility
•
Define and configure a Lab Shipping Configuration entry for each Collection
facility laboratory. This configuration determines how the LAB PENDING
ORDERS file (#69.6) data is created from the HL7 message containing the
electronic Shipping Manifest, what host facility lab tests are ordered, the
collection sample, and lab test urgency.
NOTES:
The LEDI II software application allows the host facility laboratory to accession and
use the collecting facility’s laboratory unique identifier (UID) bar-coded specimen
labels. If the host facility laboratory does not want to re-label specimens, the
collection facility laboratory must be assigned a two character NUMERIC
IDENTIFIER for the accession area that the collection facility will use for shipping
the specimens.
The host facility must not use this NUMERIC IDENTIFIER for any local
accessioning. This NUMERIC IDENTIFIER is to be used ONLY by the collection
facility laboratory for which it was assigned.
Collection Facility
NOTE: The collection and host facility laboratories must establish which lab tests
will be processed.
LIM (collection facility): The host facility laboratory must provide the collection
facility laboratory with the following information requirements:
•
Provide a list of the NLT codes and specimen requirements used by the host
facility laboratory. The Electronic Catalog Menu [LAS7 CATALOG MENU]
can be used to produce a list of the host facility NLT codes. This menu is
located on the Lab Shipping Management Menu [LA7S MGR MENU] located
on the Lab Liaison Menu [LRLIAISON]. (VA Host facility only)
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
31
LEDI II Implementation Instructions
Step #4 continu for COLLECTION facility
NOTE: The collection facility may utilize existing lab test entries in the
LABORATORY TEST file (#60), providing the entries are compatible with the host
facility’s requirements.
•
The collection facility must create a Load List to define and accept lab test
results transmitted by the host facility. The Load List must contain all of the
lab tests that are processed by the host facility laboratory. The host facility can
generate a list of their facility lab tests by using the Electronic Catalog Menu
[LA7S CATALOG MENU].
NOTE: If the host facility will be forgoing relabelling specimens received from the
collection facility then the host facility must provide the collection facility with a
NUMERIC IDENTIFIER for creating an accession area.
•
The collection facility must create an accession area in the ACCESSION file
(#68).
LIM (collection facility): Using VA FileMan [DIUSER], Enter or Edit
[DIEDIT] option assign the NUMERIC IDENTIFIER provided by the host
facility to ACCESSION file (#68), NUMERIC IDENTIFIER field (#.4). These
specimens/lab tests must be accessioned to that specific accession area.
NOTE: If the host facility will be re-labeling the collection facility specimens, then
the collection facility may use any accession area for these specimens/lab tests.
32
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
Step #5 LIM (collection facility): Lab Shipping Files Setup
Collection Facility:
Collection facility laboratory setting up a host facility laboratory for lab test requests
and shipping specimens must configure the following four Lab Shipping files in the
order listed below:
1. LAB SHIPPING METHOD file (#62.92) contains the transport method used to
ship specimens (i.e., by Courier, Taxi, FEDEX, UPS, etc.).
2. LAB SHIPPING CONDITION file (#62.93) contains entries that describe the
conditions under which a lab shipment is transported (i.e., Ambient temperature,
Frozen, Refrigerated, etc.).
3. LAB SHIPPING CONTAINER file (#62.91) contains the type of containers that
the laboratory uses to ship lab test specimens. There are basically three types of
containers used for shipping lab test specimens:
•
Primary – specimen is shipped in the original collection container.
•
Aliquot - specimen transferred to a tube/jar before shipment.
•
Packaging – packaging containers used for holding the specimen
containers.
4. LAB SHIPPING CONFIGURATION (#62.9) contains the specimen volume,
weight, collection end date/time (collection duration), patient height, and weight.
LOINC codes are used to identify patient height, weight, and specimen weight when
deemed appropriate.
NOTE: Online help is available by entering two question marks (“??”) at the field
prompt.
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
33
LEDI II Implementation Instructions
Step #5 continue for COLLECTION facility
Collection Facility:
The LAB SHIPPING METHOD file (#62.92) must be configured first. This file
contains the transport method used to ship specimens (i.e., by Courier, Taxi, FEDEX,
UPS, etc.).
LIM (collection facility): Use the Lab Shipping Management [LA7S MGR
MENU] menu, Edit Shipping Method [LA7S EDIT 62.92] option, located on the Lab
Liaison menu [LRLIAISON] to configure the LAB SHIPPING METHOD file
(#62.92).
Example: CME Edit Shipping Method [LA7S EDIT 62.92] option
Select Lab liaison menu Option: SMGR<RET>
Lab Shipping Management Menu
Select Lab Shipping Management Menu Option: ?<RET>
CFE
CTE
CME
CDE
LSU
CAT
Edit Shipping Configuration
Edit Shipping Container
Edit Shipping Method
Edit Shipping Condition
LEDI Setup
Electronic Catalog Menu
Enter ?? for more options, ??? for brief descriptions, ?OPTION for help text.
Select Lab Shipping Management Menu Option: CME<RET> Edit Shipping Method
Select SHIPPING METHOD: ?<RET>
Answer with LAB SHIPPING METHOD NAME
You may enter a new LAB SHIPPING METHOD, if you wish
NAME MUST BE 3-30 CHARACTERS, NOT NUMERIC OR STARTING WITH
PUNCTUATION
Select SHIPPING METHOD: FEDEX<RET>
Are you adding 'FEDEX' as a new LAB SHIPPING METHOD (the 1ST)? Y<RET> (Yes)
NAME: FEDEX//<RET>
Select Lab Shipping Management Menu Option: CME<RET>
Edit Shipping Method
Select SHIPPING METHOD: COURIER<RET>
Are you adding 'COURIER' as a new LAB SHIPPING METHOD (the 2ND)? Y<RET>
(Yes)
NAME: COURIER//<RET>
34
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
Step #5 continued for COLLECTION facility
Collection Facility:
The LAB SHIPPING CONDITION file (#62.93) is the second file to be configured.
This file contains entries that describe the conditions under which a lab shipment is
transported (i.e., Ambient temperature, Frozen, Refrigerated, etc.).
LIM (collection facility): Use the Lab Shipping Management [LA7S MGR
MENU] menu, Edit Shipping Condition [LA7S EDIT 62.93] option to configure the
LAB SHIPPING CONDITION file (#62.93).
Example: CDE Edit Shipping Condition [LA7S EDIT 62.93] option
Select Lab liaison menu Option: SMGR<RET>
Lab Shipping Management Menu
Select Lab Shipping Management Menu Option: ??<RET>
CFE
CTE
CME
CDE
LSU
CAT
Edit Shipping Configuration
Edit Shipping Container
Edit Shipping Method
Edit Shipping Condition
LEDI Setup
Electronic Catalog Menu
Select Lab Shipping Management Menu Option: CDE<RET> Edit Shipping Condition
Use this option to setup the Lab Shipping Condition file.
Select SHIPPING CONDITION: ?<RET>
Answer with LAB SHIPPING CONDITIONS NAME
You may enter a new LAB SHIPPING CONDITIONS, if you wish
NAME MUST BE 3-30 CHARACTERS, NOT NUMERIC OR STARTING WITH
PUNCTUATION
Select SHIPPING CONDITION: Room Temperature<RET>
Are you adding 'Room Temperature' as
a new LAB SHIPPING CONDITIONS (the 1ST)? No// Y<RET>
(Yes)
NAME: Room Temperature//<RET>
ABBREVIATION: RT<RET>
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
35
LEDI II Implementation Instructions
Step #5 continued for COLLECTION facility
Collection Facility:
The LAB SHIPPING CONTAINER file (#62.91) is the third file to be configured by
the collecting facility. This file contains the type of containers that the laboratory
uses to ship lab test specimens. There are basically three types of containers used for
shipping specimens:
•
•
•
Primary – specimen is shipped in the original collection container.
Aliquot - specimen transferred to a tube/jar before shipment.
Packaging – packaging containers used for holding the specimen containers.
LIM (collection facility): Use the Lab Shipping Management [LA7S MGR
MENU] menu, Edit Shipping Container [LA7S EDIT 62.91] option to configure the
LAB SHIPPING CONTAINER file (#62.93).
Example: CTE Edit Shipping Container [LA7S EDIT 62.91] option
Select Lab liaison menu Option:SMGR<RET> Lab Shipping Management Menu
Select Lab Shipping Management Menu Option: ?
CFE
CTE
CME
CDE
LSU
CAT
Edit Shipping Configuration
Edit Shipping Container
Edit Shipping Method
Edit Shipping Condition
LEDI Setup
Electronic Catalog Menu
Select Lab Shipping Management Menu Option: CTE<RET> Edit Shipping Container
Use this option to setup the Lab Shipping Container file.
Select SHIPPING CONTAINER: ?<RET>
Answer with LAB SHIPPING CONTAINER NAME
You may enter a new LAB SHIPPING CONTAINER, if you wish
NAME MUST BE 3-30 CHARACTERS, NOT NUMERIC OR STARTING WITH
PUNCTUATION
Select SHIPPING CONTAINER: Plastic Tube<RET>
Are you adding 'Plastic Tube' as
a new LAB SHIPPING CONTAINER (the 1ST)? No// Y<RET> (Yes)
NAME: Plastic Tube//<RET>
TYPE: ?<RET>
Enter what this container is used for.
Choose from:
1
PACKAGING
2
PRIMARY
3
ALIQUOT
TYPE: 3 ALIQUOT<RET>
36
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
Step #5 continue for COLLECTION facility
Collection Facility:
The LAB SHIPPING CONFIGURATION file (#62.9) is the fourth and final file to
be configured by the collection facility. When configuring this file to send
specimens/tests requests to a Commercial Reference Laboratory (i.e., non-VA
facility) it is very important that the INSTITUTION file (#4), for the non-VA facility
have the AGENCY CODE field (#95) entry defined as “OTHER”. The LEDI II
software asks for a three-letter identifier to identify this facility in lieu of the VA
station number. During development and testing of the LEDI II software with
Commercial Reference Laboratories the following three-letter identifiers were
used:
•
•
•
American Medical Laboratories – AML
Laboratory Corporation of America – LCA
Quest Diagnostics, Incorporated - QDI
Commercial Reference Laboratory (i.e., non-VA facility) Setup Example
LIM (collection facility): Use the Lab Shipping Management [LA7S MGR
MENU] menu, Edit Shipping Configuration [LA7S EDIT 62.9] option to configure
the LAB SHIPPING CONFIGURATION file (#62.9) for a Commercial Reference
Laboratory (i.e., non-VA facility).
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option
Select Lab Shipping Management Menu Option: ?<RET>
CFE
CTE
CME
CDE
LSU
CAT
Edit Shipping Configuration
Edit Shipping Container
Edit Shipping Method
Edit Shipping Condition
LEDI Setup
Electronic Catalog Menu ...
Select Lab Shipping Management Menu Option: CFE<RET> Edit Shipping
Configuration
Use this option to setup your Lab Shipping Configuration file.
Select OPTION NAME:
LA7S EDIT 62.9<RET>
Edit Shipping Configuration
February 2002
Edit Shipping Configuration
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
37
LEDI II Implementation Instructions
Step #5 continues for collection facility
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option used to setup
a Commercial Reference Laboratory (non-VA facility).
Select SHIPPING CONFIGURATION: XYZ COMMERCIAL REFERENCE LAB<RET>
Are you adding 'XYZ COMMERCIAL REFERENCE LAB' as a new LAB SHIPPING
CONFIGURATION (the 10TH)? No// Y<RET> (Yes)
LAB SHIPPING CONFIGURATION COLLECTING FACILITY: MILWAUKEE, WI<RET> WI
695 LAB SHIPPING CONFIGURATION HOST FACILITY: XYZ REFERENCE LAB<RET>
VAMC
WI
Select one of the following:<RET>
1
2
Collecting facility
Host facility
Are you editing this entry as the: ??<RET>
Is this entry used by the Collecting facility to ship specimens,
or by the Host facility to accept a shipment.
This determines which fields are edited in the file.
Select one of the following:
1
2
Collecting facility
Host facility
Are you editing this entry as the: 1<RET> Collecting facility
NAME: XYZ COMMERCIAL REFERENCE LAB Replace<RET>
COLLECTING FACILITY: MILWAUKEE, WI//<RET>
COLLECTING FACILITY'S SYSTEM: ??<RET>
The name of the institution, which houses the computer system from
which the sender transmitted/sent the order.
COLLECTING FACILITY'S SYSTEM: MILWAUKEE, WI<RET>
WI
VAMC
695
HOST FACILITY: XYZ REFERENCE LAB//<RET>
HOST FACILITY'S SYSTEM: XYZ REFERENCE LAB<RET>
WI
NON-VA SYSTEM IDENTIFIER: ??<RET>
If this is used to communicate with a non-VA system,
enter the 2-3 character identifier used to name the HL7 application.
NON-VA SYSTEM IDENTIFIER: XYZ<RET>
ACCOUNT NUMBER: 1234XYZ<RET>
TEST CODING SYSTEM: ??<RET>
Answer with type of coding system to use for this configuration.
Choose from:
0
NLT
1
NON-VA
2
LOINC
38
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
Step #5 continued for COLLECTION facility
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option
TEST CODING SYSTEM: ??<RET>
If orders are being sent to a non-VA facility and the facility can not
accept VA test order codes then answer with the type of coding system.
"NON-VA" indicates that the other system is using a local coding system.
The laboratory shipping software will then use the non-VA test codes
entered for each test on this configuration.
If the non-VA facility can accept VA test codes then answer "NLT" and
the software will sent VA test order codes.
VA test order codes are usually NLT codes but in the future will
Probably be LOINC codes. Selecting "LOINC" is currently not supported.
Support willbe added in a future version of the LEDI software when
LOINC coding has been implemented within VA facilities.
Choose from:
0
NLT
1
NON-VA
2
LOINC
TEST CODING SYSTEM: 1<RET> NON-VA
STATUS: ??<RET>
This field is used to designate whether this shipping configuration is
"active", i.e. selectable by the user for use in building, processing
and receipting shipments of laboratory test.
Choose from:
0
INACTIVE
1
ACTIVE
STATUS: 1<RET> ACTIVE
LAB MESSAGING LINK:<RET>
This field is used to link a shipping configuration with an
electronic transmission of shipping manifests. When a shipping
manifest is shipped, this field is checked to determine if the
software should transmit the orders on the manifest to the host
facility. It identifies the entry in the LA7 MESSAGE PARAMETER file
#62.8 to use for building and transmitting the manifest.
Note: leave this field blank when initially setting up configuration. Then
after using LEDI Setup option enter the message configuration created in file
#62.8.
LAB MESSAGING LINK:<RET>
SHIPPING METHOD: ??<RET>
Enter the method used to transport a shipment.
Answer with LAB SHIPPING METHOD NAME:
QUEST COURIER
SHIPPING METHOD: QUEST COURIER<RET>
BARCODE MANIFEST: ??<RET>
This field determines if site/patient/specimen information is barcoded
on the shipping manifest when it has a status of "shipped".
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
39
LEDI II Implementation Instructions
Step #5 continues for COLLECTION facility
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option
There are two styles of bar codes. The regular style (code="YES"),
Which was released with the original version of Laboratory Electronic
Data Interchange (LEDI), produces a long bar code. If the receiving
site, which is reading these type of bar codes, has problems then
switch to the compact style (code="YES-COMPACT"). This will produce
a shorter bar code.
Choose from:
0
NO
1
YES
2
YES-COMPACT
BARCODE MANIFEST: N<RET> NO
MANIFEST RECEIPT: ??<RET>
Allows site to have a receipt printed with a shipping manifest when
the status of the manifest is "SHIPPED". This receipt can be used to
record acknowledgment of receipt of the shipment by the courier
service used to transport the specimens to the host facility.
Choose from:
0
NO
1
YES
MANIFEST RECEIPT: Y<RET> YES
INCLUDE UNCOLLECTED SPECIMENS: ??<RET>
If specimens that are still pending receipt in lab are to build on a
shipping manifest then answer "YES". If only specimens that have been
received in the lab, i.e. have a lab arrival time, are to build then
answer "NO".
If field is blank then default is "NO".
Choose from:
0
NO
1
YES
INCLUDE UNCOLLECTED SPECIMENS: N<RET> NO
Select TEST/PROFILE: PROSTATIC SPECIFIC ANTIGEN<RET>
Are you adding 'PROSTATIC SPECIFIC ANTIGEN' as
a new TEST/PROFILE (the 1ST for this LAB SHIPPING CONFIGURATION)? No//
Y<RET> (Yes)
ACCESSION AREA: SENDOUTS// ??<RET>
COLLECTION FACILITIES:<RET>
This field is used to designate the accession area to check when
searching for tests to build onto a shipping manifest. If it is blank
then the building process will skip over this test.
HOST FACILITIES:<RET>
Not used by software at host facilities.
40
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
Step #5 continues for COLLECTION facility
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option
ACCESSION AREA: SENDOUTS//<RET>
DIVISION: ??<RET>
Collecting facilities:<RET>
If the manifest building process should only build accessions'
from a certain division on a manifest then enter the division
to screen these accessions. The division used here will be the
division associated with the user who created the accession.
This field will allow a site to screen accessions from
multiple divisions, only placing on the manifest an accession
from the specified division.
Host facilities: This field not used.
DIVISION: MILWAUKEE, WI<RET>
WI
VAMC
695
SPECIMEN: ??<RET>
This field is used to determine if a test for a particular specimen
type should build on the shipping manifest. If left blank, i.e. no
entry then all specimens for this test are eligible for building on
a shipping manifest. If a specimen type is entered then the specimen
type of the accession must match before the test is eligible for
building onto a shipping manifest.
SPECIMEN: SERUM<RET>
0X500
URGENCY: ??<RET>
COLLECTING FACILITIES:<RET>
If shipping laboratory tests that are a certain
urgency, specify the urgency that must match the
test urgency of the accession for the accession/test
to be placed on the shipping manifest.
HOST FACILITIES:<RET>
This field is used by LEDI software at the host
facility to determine the entry in the host site's
URGENCY file (#62.05) to use for ordering when the
host site has more than one entry in the URGENCY
file (#62.05) that maps to the same HL7 PRIORITY.
Example: Host site has three urgencies which
map to HL7 PRIORITY: ROUTINE (R).
Entry #
------2
9
10
February 2002
Name
--------------PATIENT WAITING
ROUTINE
NO RUSH
LEDI HL7
-------ROUTINE
ROUTINE
ROUTINE
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
41
LEDI II Implementation Instructions
Step #5 continues for COLLECTION facility
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option used to setup
a Commercial Reference Lab (non-VA facility).
If there is no mapping defined for this field,
then the LEDI software will use the last entry
in the URGENCY file that maps to HL7 PRIORITY:
ROUTINE (R); workload urgencies are excluded.
In this case the test would be ordered with a
URGENCY of NO RUSH.
If the host site enters a mapping in this field,
i.e. ROUTINE then the test will be ordered using
the host site's entry for ROUTINE in the URGENCY
file when the HL7 PRIORITY matches.
Choose from:
1
STAT
2
PATIENT WAITING
3
PRE-OP
4
CRITICAL
5
ZZADMIT
6
ZZOUTPATIENT
7
ZZPURPLE TRIANGLE
9
ROUTINE
URGENCY: 9<RET> ROUTINE
SPECIMEN CONTAINER: ??<RET>
COLLECTING FACILITIES:<RET>
The container used to hold the specimen. This could be the original
collection container or a tube/vial/jar that the specimen is
transferred to prior to shipment to another facility. It is the
container that actually hold/contains the specimen.
HOST FACILITIES:
This field is presently not used by LEDI software at the host facility.
Choose from:
Plastic Tube
Primary tube
STERILE CUP
STOOL CUP-SENDOUT
SPECIMEN CONTAINER: Primary tube<RET>
SHIPPING CONDITION: ??<RET>
COLLECTING FACILITIES:<RET>
This field describes under what temperature/environmental condition the
specimen is to be shipped. Examples would be frozen, refrigerated, or ambient
temperature.
42
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
Step #5 continues for COLLECTION facility
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option used to setup
a Commercial Reference Lab (non-VA facility).
HOST FACILITIES:<RET>
This field is presently not used by LEDI software at the
host facility.
Choose from:<RET>
Ambient/Room Temperature
FROZEN
Refrigerated
SHIPPING CONDITION: Refrigerated
PACKAGING CONTAINER: ??<RET>
COLLECTING FACILITIES:<RET>
This field is used to determine what packaging container a test's
specimen container is placed in when the specimen is being shipped
to another facility.
HOST FACILITIES:<RET>
This field is presently not used by LEDI software at the host facility.
Choose from:<RET>
Box, Container
PACKAGING CONTAINER: Box, Container<RET>
REQUIRE PATIENT HEIGHT: ??<RET>
Allows site to specify that the patient's height is sent with an order
for this test. Patient's height will be prompted for and printed on
manifest.
Choose from:<RET>
0
NO
1
YES
REQUIRE PATIENT HEIGHT: N<RET> NO
PATIENT HEIGHT UNITS: ??<RET>
Units used to measure the patient's height.
Select an entry from the LAB ELECTRONIC CODE
file (#64.061) that are measurements.
PATIENT HEIGHT UNITS:<RET>
PATIENT HEIGHT CODE: ?? <RET>
Select the appropriate LOINC code to identify the patient's height.
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
43
LEDI II Implementation Instructions
Step #5 continues for COLLECTION facility
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option used to setup
a Commercial Reference Lab (non-VA facility).
PATIENT HEIGHT CODE:<RET>
REQUIRE PATIENT WEIGHT: ??<RET>
Determines if the patient's weight is required
to be sent in the HL7 ORM order message and
printed on the shipping manifest. Actual
shipping and/or electronic transmission of a
shipping manifest will check for entry of the
patient's weight and prevent release if absent.
Choose from:
0
NO
1
YES
REQUIRE PATIENT WEIGHT:<RET>
PATIENT WEIGHT UNITS: ??<RET>
Units used to measure the patient's weight.
Select an entry from the LAB ELECTRONIC CODE
file (#64.061) that are measurements.
PATIENT WEIGHT UNITS:<RET>
PATIENT WEIGHT CODE: ??<RET>
Select the appropriate LOINC code to identify the patient's weight.
PATIENT WEIGHT CODE:<RET>
REQUIRE COLLECTION VOLUME: ??<RET>
Determines if the specimen's collection volume
is required to be sent in the HL7 ORM order
message and printed on the shipping manifest.
Actual shipping and/or electronic transmission
of a shipping manifest will check for entry of
the specimen's collection volume and prevent
release if absent.
Choose from:<RET>
0
NO
1
YES
REQUIRE COLLECTION VOLUME:<RET>
COLLECTION VOLUME UNITS: ??<RET>
Units used to measure the specimen's collection
volume. Select an entry from the LAB ELECTRONIC
CODE file #64.061) that is a measurement.
44
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
Step #5 continues for COLLECTION facility
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option used to setup
a Commercial Reference Lab (non-VA facility)
COLLECTION VOLUME UNITS:<RET>
COLLECTION VOLUME CODE: ??<RET>
Enter the appropriate LOINC code to identify
the specimen's collection volume.
COLLECTION VOLUME CODE:<RET>
REQUIRE COLLECTION END D/T: ??<RET>
Determines if the specimen's collection end
date/time is required to be sent in the HL7 ORM
order message and printed on the shipping
manifest. Actual shipping and/or electronic
transmission of a shipping manifest will check
for entry of the specimen's collection end
date/time and prevent release if absent.
Choose from:<RET>
0
NO
1
YES
REQUIRE COLLECTION END D/T:<RET>
COLLECTION DURATION UNITS: ??<RET>
Units used to calculate the specimen's
collection duration. Select an entry from the
LAB ELECTRONIC CODE file (#64.061) that is a
measurement and relate to time.
COLLECTION DURATION UNITS:<RET>
COLLECTION DURATION CODE: ??<RET>
Enter the appropriate LOINC code to identify
the specimen's collection duration.
COLLECTION DURATION CODE:<RET>
REQUIRE COLLECTION WEIGHT: ??<RET>
Determines if the specimen's collection weight
is required to be sent in the HL7 ORM order
message and printed on the shipping manifest.
Actual shipping and/or electronic transmission
of a shipping manifest will check for entry of
the specimen's collection weight and prevent
release if absent.
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
45
LEDI II Implementation Instructions
Step #5 continues for COLLECTION facility
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option used to setup
a Commercial Reference Lab (non-VA facility).
Choose from:<RET>
0
NO
1
YES
REQUIRE COLLECTION WEIGHT:<RET>
COLLECTION WEIGHT UNITS: ??<RET>
Units used to measure the specimen's collection weight.
Select an entry from the LAB ELECTRONIC CODE file
( #64.061) that is a measurement.
COLLECTION WEIGHT UNITS:<RET>
COLLECTION WEIGHT CODE: ??<RET>
Enter the appropriate LOINC code to identify
the specimen's collection weight.
COLLECTION WEIGHT CODE:<RET>
NON-VA TEST ORDER CODE: ??<RET>
Collecting faciliies:<RET>
If sending test orders to a non-VA facility use this field to store
the test order codes used by the non-VA system. It will be used when the TEST
CODING SYSTEM field (#.14) is set to "NON-VA".
Host facilities:<RET>
If receiving test orders from a non-VA collecting facility that does
not use VA NLT codes use this field to map the collecting facilities test
order codes to the corresponding VA test.
NON-VA TEST ORDER NAME:??<RET>
Collecting facility:<RET>
If sending test orders to a non-VA facility use this field to store
the test order name used by the non-VA system. The lab software will identify
the test name on the non-VA system when orders are transmitted electronically.
uses this field. It will be used when the TEST CODING SYSTEM field (#.14) is
set to "NON-VA".
Host facility: <RET>
Not used - mapping of non-VA tests coding systems
for test order codes is via NON-VA TEST ORDER CODE
field (#5.1).
NON-VA TEST ORDER CODE: XYZ-GLU123<RET>
Select TEST/PROFILE:
46
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
Step #5 continues for COLLECTION facility
LIM (collection facility): Use the Lab Shipping Management [LA7S MGR
MENU] menu, Edit Shipping Configuration [LA7S EDIT 62.9] option to configure
the LAB SHIPPING CONFIGURATION file (#62.9) for a VA Facility.
Example: CFE: Edit Shipping Configuration [LA7S EDIT 62.9] option used to
configure a VA Facility.
Select Lab Shipping Management Menu Option: ?<RET>
CFE
CTE
CME
CDE
LSU
CAT
Edit Shipping Configuration
Edit Shipping Container
Edit Shipping Method
Edit Shipping Condition
LEDI Setup
Electronic Catalog Menu ...
Select Lab Shipping Management Menu Option:CFE<RET> Edit Shipping
Configuration<RET>
Select SHIPPING CONFIGURATION:?<RET>
Answer with LAB SHIPPING CONFIGURATION NAME, or COLLECTING FACILITY, or
HOST FACILITY, or HOST FACILITY'S SYSTEM, or COLLECTING FACILITY'S SYSTEM
Do you want the entire 11-Entry LAB SHIPPING CONFIGURATION List?<RET>
You may enter a new LAB SHIPPING CONFIGURATION, if you wish
Answer must be 3-30 characters in length.
Select SHIPPING CONFIGURATION: DALLAS OIFO SENDOUTS<RET>
Are you adding 'DALLAS OIFO SENDOUTS' as
a new LAB SHIPPING CONFIGURATION (the 12TH)? No// Y<RET>
(Yes)
LAB SHIPPING CONFIGURATION COLLECTING FACILITY: ?<RET>
Select the institution which will be collecting and shipping lab
test specimens.
Answer with INSTITUTION NAME, or STATION NUMBER, or OFFICAL VA NAME, or
CURRENT LOCATION
Do you want the entire 290-Entry INSTITUTION List?<RET>
LAB SHIPPING CONFIGURATION COLLECTING FACILITY: MILWAUKEE, WI<RET> WI VAMC 695
LAB SHIPPING CONFIGURATION HOST FACILITY: ?<RET>
Select the institution which will be the Host facility receiving the lab
test shipment.
Answer with INSTITUTION NAME, or STATION NUMBER, or OFFICAL VA NAME, or
CURRENT LOCATION
Do you want the entire 290-Entry INSTITUTION List?<RET>
LAB SHIPPING CONFIGURATION HOST FACILITY: DALLAS OIFO<RET>
TX ISC 170
Select one of the following:<RET>
1
2
February 2002
Collecting facility
Host facility
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
47
LEDI II Implementation Instructions
Step #5 continues for COLLECTION facility
Example: CFE: Edit Shipping Configuration [LA7S EDIT 62.9] option
Are you editing this entry as the: ?<RET>
Is this entry used by the Collecting facility to ship specimens, or by the
Host facility to accept a shipment.
This determines which fields are edited in the file.
Select one of the following:
1
Collecting facility
2
Host facility
Are you editing this entry as the: 1<RET> Collecting facility
NAME: DALLAS OIFO SENDOUTS Replace<RET>
COLLECTING FACILITY: MILWAUKEE, WI//<RET>
COLLECTING FACILITY'S SYSTEM: ?<RET>
Institution which has collector's computer system.
Answer with INSTITUTION NAME, or STATION NUMBER, or OFFICAL VA NAME, or
CURRENT LOCATION
Do you want the entire 290-Entry INSTITUTION List?<RET>
COLLECTING FACILITY'S SYSTEM: MILWAUKEE, WI<RET>
WI
VAMC
695
HOST FACILITY: DALLAS OIFO//<RET>
HOST FACILITY'S SYSTEM: ?<RET>
Institution which has host's computer system.
Answer with INSTITUTION NAME, or STATION NUMBER, or OFFICAL VA NAME, or
CURRENT LOCATION
Do you want the entire 290-Entry INSTITUTION List?<RET>
HOST FACILITY'S SYSTEM: DALLAS OIFO<RET>
TX
ISC
170
STATUS: ? <RET>
Enter the status of this sending/receiving combination.
Choose from:
0
INACTIVE
1
ACTIVE
STATUS: A<RET> ACTIVE
LAB MESSAGING LINK:<RET>
SHIPPING METHOD: ?<RET>
Enter the method used to transport a shipment.
Answer with LAB SHIPPING METHOD NAME: QUEST COURIER<RET>
SHIPPING METHOD: QUEST COURIER<RET>
BARCODE MANIFEST: ??<RET>
This field determines if site/patient/specimen information is barcoded
on the shipping manifest when it has a status of "shipped".
There are two styles of bar codes. The regular style (code="YES"),
which was released with the original version of Laboratory Electronic
Data Interchange (LEDI), produces a long bar code. If the receiving
site, which is reading these type of bar codes, has problems then
switch to the compact style (code="YES-COMPACT"). This will produce a
shorter bar code.
48
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
Step #5 continues for the COLLECTION facility
Example: CFE: Edit Shipping Configuration [LA7S EDIT 62.9] option used to
configure a VA Facility
Choose from:?<RET>
0
NO
1
YES
2
YES-COMPACT
BARCODE MANIFEST:2<RET> YES-COMPACT
MANIFEST RECEIPT:??<RET>
Allows site to have a receipt printed with a shipping manifest when
the status of the manifest is "SHIPPED". This receipt can be used to
record acknowledgment of receipt of the shipment by the courier
service used to transport the specimens to the host facility.
Choose from:??<RET>
0
NO
1
YES
MANIFEST RECEIPT: 1<RET> YES
INCLUDE UNCOLLECTED SPECIMENS:??<RET>
If specimens that are still pending receipt in lab are to build on a
shipping manifest then answer "YES". If only specimens that have been
received in the lab, i.e. have a lab arrival time, are to build then
answer "NO".
If field is blank then default is "NO".
Choose from:<RET>
0
NO
1
YES
INCLUDE UNCOLLECTED SPECIMENS: N<RET> NO
Select TEST/PROFILE: ?<RET>
You may enter a new TEST/PROFILE, if you wish
Enter the laboratory test that is used by this shipping
configuration.
Answer with LABORATORY TEST NAME, or LOCATION (DATA NAME), or
PRINT NAME
Do you want the entire 1359-Entry LABORATORY TEST List?
Select TEST/PROFILE: THEOPHYLLINE<RET>
Are you adding 'THEOPHYLLINE' as a new TEST/PROFILE (the 1ST for this
LAB SHIPPING CONFIGURATION)? No// Y<RET> (Yes)
ACCESSION AREA: ??<RET>
COLLECTION FACILITIES:<RET>
This field is used to designate the accession area to check when
searching for tests to build onto a shipping manifest. If it is blank
then the building process will skip over this test.
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
49
LEDI II Implementation Instructions
Step #5 continues for COLLECTION facility
Example: CFE: Edit Shipping Configuration [LA7S EDIT 62.9] option used to
configure a VA Facility.
HOST FACILITIES:<RET>
Not used by software at host facilities.
ACCESSION AREA: SENDOUTS<RET>
DIVISION: ?<RET>
Enter the accession's division (?? - additional help).
Answer with INSTITUTION NAME, or STATION NUMBER, or OFFICAL VA NAME, or
CURRENT LOCATION
Do you want the entire 290-Entry INSTITUTION List?<RET>
DIVISION: ??<RET>
Collecting facilities:<RET>
If the manifest building process should only build accessions'
from a certain division on a manifest then enter the division
to screen these accessions. The division used here will be the
division associated with the user who created the accession.
This field will allow a site to screen accessions from
multiple divisions, only placing on the manifest an accession
from the specified division.
Host facilities: This field not used.
DIVISION: MILWAUKEE, WI
WI
VAMC
695
SPECIMEN: ??<RET>
This field is used to determine if a test for a particular specimen
type should build on the shipping manifest. If left blank, i.e. no
entry then all specimens for this test are eligible for building on
a shipping manifest. If a specimen type is entered then the specimen
type of the accession must match before the test is eligible for
building onto a shipping manifest.
SPECIMEN: SERUM<RET>
0X500
URGENCY: ?? <RET>
COLLECTING FACILITIES:<RET>
If shipping laboratory tests that are a certain
urgency, specify the urgency that must match the
test urgency of the accession for the accession/test
to be placed on the shipping manifest.
50
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
Step #5 continues for the COLLECTION facility
Example: CFE: Edit Shipping Configuration [LA7S EDIT 62.9] option used to
configure a VA Facility
HOST FACILITIES:<RET>
This field is used by LEDI software at the host
facility to determine the entry in the host site's
URGENCY file (#62.05) to use for ordering when the
host site has more than one entry in the URGENCY
file (#62.05) that maps to the same HL7 PRIORITY.
Example: Host site has three urgencies which map to HL7 PRIORITY:
ROUTINE (R).
Entry #
------2
9
10
Name
--------------PATIENT WAITING
ROUTINE
NO RUSH
LEDI HL7
-------ROUTINE
ROUTINE
ROUTINE
If there is no mapping defined for this field,
then the LEDI software will use the last entry
in the URGENCY file that maps to HL7 PRIORITY:
ROUTINE (R); workload urgencies are excluded.
In this case the test would be ordered with a
URGENCY of NO RUSH.
If the host site enters a mapping in this field,
i.e. ROUTINE then the test will be ordered using
the host site's entry for ROUTINE in the URGENCY
file when the HL7 PRIORITY matches.
Choose from:
1
STAT
2
PATIENT WAITING
3
PRE-OP
4
CRITICAL
5
ADMIT
6
OUTPATIENT
7
PURPLE TRIANGLE
9
ROUTINE
URGENCY:9<RET>ROUTINE
SPECIMEN CONTAINER:??<RET>
COLLECTING FACILITIES:<RET>
The container used to hold the specimen. This could be the original
collection container or a tube/vial/jar that the specimen is
transferred to prior to shipment to another facility. It is the
container that actually hold/contains the specimen.
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
51
LEDI II Implementation Instructions
Step #5 continues for COLLECTION facility
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option used to
configure a VA Facility.
HOST FACILITIES:<RET>
This field is presently not used by LEDI software at the host
facility.
SPECIMEN CONTAINER: Primary tube<RET>
SHIPPING CONDITION: ??<RET>
COLLECTING FACILITIES:<RET>
This field describes under what temperature/environmental condition
the specimen is to be shipped. Examples would be frozen, refrigerated
or ambient temperature.
HOST FACILITIES:<RET>
This field is presently not used by LEDI software at the
host facility.
SHIPPING CONDITION: Refrigerated<RET>
PACKAGING CONTAINER: ??<RET>
COLLECTING FACILITIES: <RET>
This field is used to determine what packaging container a test's
specimen container is placed in when the specimen is being shipped
to another facility.
HOST FACILITIES:<RET>
This field is presently not used by LEDI software at the
host facility.
PACKAGING CONTAINER: Box, Container<RET>
REQUIRE PATIENT HEIGHT: ?? <RET>
Allows site to specify that the patient's height is sent with
an order for this test. Patient's height will be prompted for
and printed on manifest.
Choose from: <RET>
0
NO
1
YES
REQUIRE PATIENT HEIGHT: n<RET> NO
PATIENT HEIGHT UNITS: ??<RET>
Units used to measure the patient's height.
Select an entry from the LAB ELECTRONIC CODE
file (#64.061) that are measurements.
52
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
Step #5 continues for COLLECTION facility
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option used to
configure a VA Facility.
PATIENT HEIGHT UNITS: <RET>
PATIENT HEIGHT CODE: ??<RET>
Select the appropriate LOINC code to identify
the patient's height.
PATIENT HEIGHT CODE:<RET>
REQUIRE PATIENT WEIGHT: YES// ??<RET>
Determines if the patient's weight is required
to be sent in the HL7 ORM order message and
printed on the shipping manifest. Actual
shipping and/or electronic transmission of a
shipping manifest will check for entry of the
patient's weight and prevent release if absent.
Choose from:<RET>
0
NO
1
YES
REQUIRE PATIENT WEIGHT: YES//<RET>
PATIENT WEIGHT UNITS: kg kg
MEASUREMENTS
Kilograms
PATIENT WEIGHT CODE: ??<RET>
Select the appropriate LOINC code to identify
the patient's weight.
^<RET>
PATIENT WEIGHT CODE: 3142<RET>
-7
BODY WEIGHT:MASS:PT:~PATIENT:QN:STATED
REQUIRE COLLECTION VOLUME: ??<RET>
Determines if the specimen's collection volume
is required to be sent in the HL7 ORM order
message and printed on the shipping manifest.
Actual shipping and/or electronic transmission
of a shipping manifest will check for entry of
the specimen's collection volume and prevent
release if absent.
Choose from:<RET>
0
NO
1
YES
REQUIRE COLLECTION VOLUME: n<RET> NO
COLLECTION VOLUME UNITS: ??<RET>
Units used to measure the specimen's collection
volume. Select an entry from the LAB ELECTRONIC
CODE file (#64.061) that is a measurement.
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
53
LEDI II Implementation Instructions
Step #5 continues for COLLECTION facility
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option used to
configure a VA Facility.
COLLECTION VOLUME UNITS:<RET>
COLLECTION VOLUME CODE: ??<RET>
Enter the appropriate LOINC code to identify
the specimen's collection volume.
COLLECTION VOLUME CODE:<RET>
REQUIRE COLLECTION END D/T: ??<RET>
Determines if the specimen's collection end
date/time is required to be sent in the HL7 ORM
order message and printed on the shipping
manifest. Actual shipping and/or electronic
transmission of a shipping manifest will check
for entry of the specimen's collection end
date/time and prevent release if absent.
Choose from:<RET>
0
NO
1
YES
REQUIRE COLLECTION END D/T: n<RET> NO
COLLECTION DURATION UNITS: ??<RET>
Units used to calculate the specimen's
collection duration. Select an entry from the
LAB ELECTRONIC CODE file (#64.061) that is a
measurement and relate to time.
COLLECTION DURATION UNITS:<RET>
COLLECTION DURATION CODE: ??<RET>
Enter the appropriate LOINC code to identify the specimen's collection
duration.
54
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
Step #5 continues for COLLECTION facility
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option used to
configure a VA Facility
COLLECTION DURATION CODE:<RET>
REQUIRE COLLECTION WEIGHT: ??<RET>
Determines if the specimen's collection weight
is required to be sent in the HL7 ORM order
message and printed on the shipping manifest.
Actual shipping and/or electronic transmission
of a shipping manifest will check for entry of
the specimen's collection weight and prevent
release if absent.
Choose from:<RET>
0
NO
1
YES
REQUIRE COLLECTION WEIGHT: n<RET> NO
COLLECTION WEIGHT UNITS: ??<RET>
Units used to measure the specimen's collection weight.
Select an entry from the LAB ELECTRONIC CODE file
( #64.061) that is a measurement.
COLLECTION WEIGHT UNITS:<RET>
COLLECTION WEIGHT CODE: ??<RET>
Enter the appropriate LOINC code to identify
the specimen's collection weight.
COLLECTION WEIGHT CODE:<RET>
Select TEST/PROFILE:<RET>
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
55
LEDI II Implementation Instructions
Step #6 LIM (host facility): LAB SHIPPING CONFIGURATION file (#62.9)
Setup
Host Facility:
Host facility laboratory setting up a collection facility laboratory to receive electronic
orders must configure ONLY the LAB SHIPPING CONFIGURATION file (#62.9).
This file is used to define the two members (institutions) that have a relationship.
File #62.9 groups both facilities (collection/host facilities), describes the specimen ID
used, types of tests/specimens being shipped, identifies the collection/host facility, if
the collection/host facilities are linked electronically, and if the collection/host
facilities are on the same computer system. Additonally, the LAB SHIPPING
CONFIGURATION file (#62.9) allows the host facility to map local tests, urgencies,
and specimen types to incoming orders from the collection facilities. This mapping
enables the appropriate host facitily test to be ordered when the specimen shipment
arrives at the host facility.
Commercial Reference Laboratory (non-VA facility) Setup
LIM (host facility): Select the Lab Shipping Management [LA7S MGR MENU]
menu, Edit Shipping Configuration [LA7S EDIT 62.9] option to configure the LAB
SHIPPING CONFIGURATION file (#62.9) for a Commercial Reference
Laboratory (non-VA facility).
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option
Select Lab Shipping Management Menu Option:?<RET>
CFE
CTE
CME
CDE
LSU
CAT
Edit Shipping Configuration
Edit Shipping Container
Edit Shipping Method
Edit Shipping Condition
LEDI Setup
Electronic Catalog Menu ...
Select Lab Shipping Management Menu Option:CFE<RET> Edit Shipping
Configuration
Select SHIPPING CONFIGURATION:XYZ COLLECTING LAB<RET>
Are you adding 'XYZ COLLECTING LAB' as
a new LAB SHIPPING CONFIGURATION (the 11TH)? No//Y<RET> (Yes)
LAB SHIPPING CONFIGURATION COLLECTING FACILITY:XYZ<RET>REFERENCE LAB WI
LAB SHIPPING CONFIGURATION HOST FACILITY:MILWAUKEE<RET> WI VAMC 695
56
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
Step #6 continues for HOST facility
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option is used to
configure a Commercial Reference Laboratory (non-VA facility).
Select one of the following:<RET>
1
Collecting facility
2
Host facility
Are you editing this entry as the: 2<RET>
Host facility
Select one of the following: <RET>
0
1
2
Do NOT copy
Another Shipping Configuration
Test Catalog - LABORATORY TEST File #60
Copy a test profile from: Do NOT copy//<RET>
NAME: XYZ COLLECTING LAB//<RET>
COLLECTING FACILITY: XYZ REFERENCE LAB//<RET>
COLLECTING FACILITY'S SYSTEM:XYZ REFERENCE LAB<RET>
WI
HOST FACILITY: MILWAUKEE, WI//<RET>
HOST FACILITY'S SYSTEM: MILWAUKEE,WI MILWAUKEE, WI WI VAMC
695<RET>
NON-VA SYSTEM IDENTIFIER: ??<RET>
If this is used to communicate with a non-VA system, enter the 2-3
character identifier used to name the HL7 application.
NON-VA SYSTEM IDENTIFIER: XYZ<RET>
TEST CODING SYSTEM: ?<RET>
Answer with type of coding system to use for this configuration.
Choose from:
0
NLT
1
NON-VA
2
LOINC
TEST CODING SYSTEM: 1<RET> NON-VA
SPECIMEN CODING SYSTEM: ?<RET>
Answer with type of coding system to use for this configuration.
Choose from:
0
HL7 TABLE 0070
1
LOCAL-NON HL7
SPECIMEN CODING SYSTEM:??<RET>
If orders are received from a non-VA facility and the facility can
not transmit HL7 specimen codes from HL7 table 0070 then answer
with the type of coding system "LOCAL".
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
57
LEDI II Implementation Instructions
Step #6 continues for HOST facility
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option is used to
configure a Commercial Reference Laboratory (non-VA facility).
Choose from: <RET>
0
HL7 TABLE 0070
1
LOCAL-NON HL7
SPECIMEN CODING SYSTEM: 1 <RET> LOCAL-NON HL7
STATUS: ??
This field is used to designate whether this shipping configuration is
"active", i.e. selectable by the user for use in building, processing
and receipting shipments of laboratory test.
Choose from:
0
INACTIVE
1
ACTIVE
STATUS: A <RET>ACTIVE
COLLECTING FACILITY'S SPEC ID: ??<RET>
This field is used to determine the source of the specimen id
(Lab UID) used to identify a specimen. It is used by the
Host facility when accessioning tests requests from Collecting
facilities to determine if the Host facility needs to generate
a new specimen id/label or use the specimen id/label from the
Collecting facility.
If both the Collecting and Host facilities on operating on
the same computer system then select #3 - SAME SYSTEM.
Choose from:
0
COLLECTING FACILITY
1
HOST FACILTY
3
SAME SYSTEM
COLLECTING FACILITY'S SPEC ID: 0<RET> COLLECTING FACILITY
LAB MESSAGING LINK:<RET>
Select TEST/PROFILE:
PROSTATIC SPECIFIC ANTIGEN<RET>
Are you adding 'PROSTATIC SPECIFIC ANTIGEN' as
a new TEST/PROFILE (the 1ST for this LAB SHIPPING CONFIGURATION)? No//
Y<RET> (Yes)
TEST/PROFILE ACCESSION AREA: CHEMISTRY
1
CHEMISTRY
2
CHEMISTRY MISC
CHOOSE 1-2: 1<RET> CHEMISTRY
TEST/PROFILE SPECIMEN: SERUM
0X500
URGENCY: ??<RET>
COLLECTING FACILITIES:<RET>
If shipping laboratory tests that are a certain
urgency, specify the urgency that must match the
test urgency of the accession for the accession/test
to be placed on the shipping manifest.
58
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
Step #6 continued for HOST facility
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option is used to
configure a Commercial Reference Laboratory (non-VA facility).
HOST FACILITIES:<RET>
This field is used by LEDI software at the host
facility to determine the entry in the host site's
URGENCY file (#62.05) to use for ordering when the
host site has more than one entry in the URGENCY
file (#62.05) that maps to the same HL7 PRIORITY.
Example: Host site has three urgencies which
map to HL7 PRIORITY: ROUTINE (R).
Entry #
------2
9
10
Name
--------------PATIENT WAITING
ROUTINE
NO RUSH
LEDI HL7
-------ROUTINE
ROUTINE
ROUTINE
If there is no mapping defined for this field,
then the LEDI software will use the last entry
in the URGENCY file that maps to HL7 PRIORITY:
ROUTINE (R); workload urgencies are excluded.
In this case the test would be ordered with a
URGENCY of NO RUSH.
If the host site enters a mapping in this field,
i.e. ROUTINE then the test will be ordered using
the host site's entry for ROUTINE in the URGENCY
file when the HL7 PRIORITY matches.
Choose from:<RET>
1
STAT
2
PATIENT WAITING
3
PRE-OP
4
CRITICAL
5
ZZADMIT
6
ZZOUTPATIENT
7
ZZPURPLE TRIANGLE
9
ROUTINE
URGENCY:9<RET>ROUTINE
HOST COLLECTION SAMPLE:SERUM??<RET>
Enter the collection sample to be associated with this test.
HOST COLLECTION SAMPLE:??<RET>
COLLECTING FACILITIES:<RET>
This field is presently not used by LEDI software at the collecting facility.
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
59
LEDI II Implementation Instructions
Step #6 continues for HOST facility
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option is used to
configure a Commercial Reference Laboratory (non-VA facility).
HOST FACILITIES:<RET>
This field is used by host facility laboratories to designate the
collection sample to be used when processing electronic orders for
this test/specimen combination. It is used to build the order in
file # 69.6, LAB PENDING ORDERS, with the test/specimen/collection
sample that the host facility uses.
HOST COLLECTION SAMPLE: BLOOD<RET>
NON-VA TEST ORDER CODE: PSA123//??<RET>
Collecting faciliies:
If sending test orders to a non-VA facility use this field to store
the test order codes used by the non-VA system. It will be used when the TEST
CODING SYSTEM field (#.14) is set to "NON-VA".
Host facilities:<RET>
If receiving test orders from a non-VA collecting facility that does
not use VA NLT codes use this field to map the collecting facilities test
order codes to the corresponding VA test.
NON-VA TEST ORDER CODE: PSA123//<RET>
NON-VA TEST ORDER NAME: PSA// ??<RET>
Collecting facility:
If sending test orders to a non-VA facility use this field to store
the test order name used by the non-VA system. The lab software will identify
the test name on the non-VA system when orders are transmitted electronically.
It will be used when the TEST CODING SYSTEM field (#.14) is set to "NON-VA".
Host facility:<RET>
Not used - mapping of non-VA tests coding systems
for test order codes is via NON-VA TEST ORDER CODE
field (#5.1).
NON-VA TEST ORDER NAME: PSA//<RET>
NON-VA SPECIMEN CODE: SERUM//??<RET>
Collecting facilities: not applicable
60
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
Step #6 continues for HOST facility
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option is used to
configure a Commercial Reference Laboratory (non-VA facility).
Host facilities:<RET>
If receiving test orders from a non-VA facility that
does not used HL7 Table 0070 Specimen Source this
field is used to store the specimen name used by the
non-VA system. It is based on the value of field
SPECIMEN CODING SYSTEM (#.15) and only used if
LOCAL-NON HL7 is selected.
NON-VA SPECIMEN CODE: SERUM//<RET>
NON-VA SPECIMEN NAME: SERUM//??<RET>
Collecting facilities: not applicable
Host facilities:
If receiving test orders from a non-VA facility that
does not used HL7 Table 0070 Specimen Source this
field is used to store the specimen name used by the
non-VA system. It is based on the value of field
SPECIMEN CODING SYSTEM (#.15) and only used if
LOCAL-NON HL7 is selected.
NON-VA SPECIMEN NAME: SERUM<RET>
Select TEST/PROFILE: <RET>
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
61
LEDI II Implementation Instructions
Step #6 continues for HOST facility
VA Facility Setup
LIM (host facility): Use the Lab Shipping Management [LA7S MGR MENU]
menu, Edit Shipping Configuration [LA7S EDIT 62.9] option to configure the LAB
SHIPPING CONFIGURATION file (#62.9) for a VA facility.
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option
Select Lab Shipping Management Menu Option: Edit Shipping Configuration<RET>
Select SHIPPING CONFIGURATION:DALLAS OIFO COLLECTING<RET>
Are you adding 'DALLAS OIFO COLLECTING' as
a new LAB SHIPPING CONFIGURATION (the 13TH)? No//Y<RET> (Yes)
LAB SHIPPING CONFIGURATION COLLECTING FACILITY:?<RET>
Select the institution which will be collecting and shipping lab test
specimens.
Answer with INSTITUTION NAME, or STATION NUMBER, or OFFICAL VA NAME, or
CURRENT LOCATION
Do you want the entire 290-Entry INSTITUTION List?<RET>
LAB SHIPPING CONFIGURATION COLLECTING FACILITY:DALLAS OIFO TX ISC 170
LAB SHIPPING CONFIGURATION HOST FACILITY:MILWAUKEE, WI
Select one of the following:<RET>
1
Collecting facility
2
Host facility
Are you editing this entry as the:2<RET>
Host facility
Select one of the following:<RET>
0
1
2
Do NOT copy
Another Shipping Configuration
Test Catalog - LABORATORY TEST File #60
Copy a test profile from: Do NOT copy//<RET>
NAME: DALLAS OIFO COLLECTING Replace<RET>
COLLECTING FACILITY: DALLAS OIFO//<RET>
COLLECTING FACILITY'S SYSTEM: DALLAS OIFO
TX
ISC
170
HOST FACILITY: MILWAUKEE, WI//<RET>
HOST FACILITY'S SYSTEM: ? <RET>
Institution which has host's computer system.
Answer with INSTITUTION NAME, or STATION NUMBER, or OFFICAL VA NAME, or
CURRENT LOCATION
Do you want the entire 290-Entry INSTITUTION List?<RET>
HOST FACILITY'S SYSTEM: MILWAUKEE, WI<RET>
WI
VAMC
695
STATUS: A<RET>ACTIVE
COLLECTING FACILITY'S SPEC ID:?<RET>
Select the source of the specimen id provided by the collector.
62
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
Step #6 continues for HOST facility
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option is used to
configure a VA facility.
Choose from:
0
COLLECTING FACILITY
1
HOST FACILTY
3
SAME SYSTEM
COLLECTING FACILITY'S SPEC ID: 0<RET> COLLECTING FACILITY
Select TEST/PROFILE: THEOPHYLLINE<RET>
Are you adding 'THEOPHYLLINE' as a new TEST/PROFILE (the 1ST for this
LAB SHIPPING CONFIGURATION)? No// Y (Yes)
URGENCY: ?
Collecting facility - if only certain urgencies are shipped, specify the
accession's test urgency. Host facility - see field description (??) for
information.
Select regular urgencies only. Workload urgencies are excluded.
Answer with URGENCY, or NUMBER
Do you want the entire URGENCY List?<RET>
URGENCY: ??<RET>
COLLECTING FACILITIES:
If shipping laboratory tests that are a certain
urgency, specify the urgency that must match the
test urgency of the accession for the accession/test
to be placed on the shipping manifest.
HOST FACILITIES:
This field is used by LEDI software at the host
facility to determine the entry in the host site's
URGENCY file (#62.05) to use for ordering when the
host site has more than one entry in the URGENCY
file (#62.05) that maps to the same HL7 PRIORITY.
Example: Host site has three urgencies which
map to HL7 PRIORITY: ROUTINE (R).
Entry #
------2
9
10
Name
--------------PATIENT WAITING
ROUTINE
NO RUSH
LEDI HL7
-------ROUTINE
ROUTINE
ROUTINE
If there is no mapping defined for this field,
then the LEDI software will use the last entry
in the URGENCY file that maps to HL7 PRIORITY:
ROUTINE (R); workload urgencies are excluded.
In this case the test would be ordered with a
URGENCY of NO RUSH.
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
63
LEDI II Implementation Instructions
Step #6 continues for HOST facility
Example: CFE Edit Shipping Configuration [LA7S EDIT 62.9] option is used to
configure a VA facility.
If the host site enters a mapping in this field,
i.e. ROUTINE then the test will be ordered using
the host site's entry for ROUTINE in the URGENCY
file when the HL7 PRIORITY matches.
Choose from:<RET>
1
STAT
2
PATIENT WAITING
3
PRE-OP
4
CRITICAL
5
ADMIT
6
OUTPATIENT
7
PURPLE TRIANGLE
9
ROUTINE
URGENCY: ROUTINE
HOST COLLECTION SAMPLE: ??<RET>
COLLECTING FACILITIES:<RET>
This field is presently not used by LEDI software at the collecting
facility.
HOST FACILITIES:<RET>
This field is used by host facility laboratories to designate
the collection sample to be used when processing electronic orders
for this test/specimen combination. It is used to build the order
in file # 69.6, LAB PENDING ORDERS, with the test/specimen/
collection sample that the host facility uses.
HOST COLLECTION SAMPLE: SERUM??<RET>
Enter the collection sample to be associated with this test.
HOST COLLECTION SAMPLE: BLOOD
1
BLOOD
BLOOD
CULTURE SET
2
BLOOD
BLOOD
GREEN
3
BLOOD
SERUM
RED
4
BLOOD
PLASMA
GREEN
5
BLOOD
PLASMA
LAVEN W/DYE
Press <RETURN> to see more, '^' to exit this list, OR
CHOOSE 1-5: 3<RET> BLOOD
SERUM
RED
Select TEST/PROFILE:<RET>
64
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
Step #7 LIM: LEDI II Setup for Laboratory Auto-Instruments and HL7
Environment
The LEDI Setup [LA7V SETUP] option allows the LIM to setup a collection and/or
host facility laboratory Auto-Instruments and HL7 environment. This option is
located on the Lab Shipping Management [LA7S MGR MENU] menu of the Lab
Liaison [LRLIAISON] menu.
The LEDI software application is using the INSTITUTION file (#4), DOMAIN field
(#60), to route HL7 messages to the collection and host facilities when VA MailMan is
used as the HL7 transport protocol.
The collection facility’s STATION NUMBER must be defined in the host facility
INSTITUTION file (#4), DOMAIN field (#60), when VA MailMan is used as the HL7
transport protocol.
The host facility’s STATION NUMBER must be defined in the collection facility
INSTITUTION file (#4), DOMAIN field (#60), when VA MailMan is used as the HL7
transport protocol.
NOTES:
Multi-divisional facilities should ONLY use the LEDI Setup option for the primary
facility. All other facilities within a multi-divisional facility will use the primary
facility to transmit and receive HL7 messages.
If the collecting facility is a division of a multi-divisional facility then setup the
primary facility’s system as the collecting facility.
If the host facility is a multi-divisional facility then setup the primary facility’s
system as the host facility.
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
65
LEDI II Implementation Instructions
Step #7 continues - LEDI II Setup for Laboratory Auto-Instruments and
HL7 Environment
NOTE: The two-instution names used in the following screen displays are examples
ONLY. Do NOT use these two institutions host or collection facility.
Example: LSU LEDI Setup
Select Lab liaison menu Option: SMGR <RET> Lab Shipping Management Menu
Select Lab Shipping Management Menu Options:?<RET>
CFE
CTE
CME
CDE
LSU
CAT
Edit Shipping Configuration
Edit Shipping Container
Edit Shipping Method
Edit Shipping Condition
LEDI Setup
Electronic Catalog Menu
Select Lab Shipping Management Menu Option:LSU<RET>
LEDI Setup
-----------------------------------------------------------------------------LEDI Setup
-----------------------------------------------------------------------------COLLECTION Labs:
HOST Labs
:
1.
2.
Use option #1 to setup HOST labs.
Use option #2 to setup COLLECTION labs.
Add/Edit HOST Lab
Add/Edit COLLECTION Lab
Enter a number (1-2):?<RET>
Option #1 will setup HOST site auto-instruments, HOST site message
configuration, and HOST and COLLECTION sites HL7 environment.
Option #2 will setup COLLECTION site auto-instruments, COLLECTION site message
configuration, and COLLECTION and HOST sites HL7 environment.
Option #1 and #2 SHOULD be used by sites that are both a HOST
and a COLLECTION site.
Enter a number (1-2):1<RET>
66
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
Step #7 continues
---------------------------------------------------------------------------HOST Lab(s)
---------------------------------------------------------------------------1.
2.
3.
4.
5.
DALLAS CIOFO (LA7V HOST 170)
BIRMINGHAM, AL. (LA7V HOST 521)
HINES, IL (LA7V HOST 578)
MILWAUKEE, WI (LA7V HOST 695)
Add HOST Lab
Enter a number (1-5): ? <RET>
Enter a '5' to create a new HOST lab.
Enter a number between 1 and 5.
Enter a number (1-5): 5<RET>
---------------------------------------------------------------------------HOST Lab Setup
---------------------------------------------------------------------------1 1. HOST Lab: <RET>
2. Logical Link (MailMan or TCP/IP): <RET>
3. Auto Instrument: <RET>
Enter a number (1-3): ?<RET>
Enter a '1' to create the HL7 environment along with the Auto-Instrument
and LA7 Message Configuration.
Enter a '2' to create the link between the HOST and COLLECTION labs.
Enter a '3' to identify the list of test you expect back from the HOST lab.
Enter a '4' to configure the LA7 MESSAGE PARAMETER file.
Enter a '5' to identify the type of Error Conditions you wish to receive for
this Auto-Instrument.
Enter a number (1-3): 1<RET>
Select INSTITUTION NAME: [Type your host laboratory’s institution name]<RET>
Select INSTITUTION NAME: ?<RET>
Answer with INSTITUTION NAME, or STATION NUMBER, or OFFICAL VA NAME, or
CURRENT LOCATION
Do you want the entire 289-Entry INSTITUTION List? <RET>
Select INSTITUTION NAME: XYZ REFERENCE LAB
WI<RET>
Enter a non-VA code (2-3 alpha character): XYZ<RET>
Select LAB SHIPPING CONFIGURATION NAME: XYZ REFERENCE LAB<RET> WI XYZ
COLLECTING LAB
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
67
LEDI II Implementation Instructions
Step #7 continues
XYZ^MILWAUKEE, WI
Setting up the following Host Labs for MILWAUKEE, WI
Updating HL7 APPLICATION PARAMETER file (#771).
Adding LA7V HOST XYZ
Updating PROTOCOL file (#101).
LA7V Receive Results from XYZ
Located in the LA7 (LAB UNIVERSAL INTERFACE) namespace.
Adding LA7V Receive Results from XYZ
LA7V Process Results from XYZ
Located in the LA7 (LAB UNIVERSAL INTERFACE) namespace.
Adding LA7V Process Results from XYZ
LA7V Order to XYZ
Located in the LA7 (LAB UNIVERSAL INTERFACE) namespace.
Adding LA7V Order to XYZ
LA7V Send Order to XYZ
Located in the LA7 (LAB UNIVERSAL INTERFACE) namespace.
Adding LA7V Send Order to XYZ
Updating LA7 MESSAGE PARAMETER file (#62.48) for the HOST Lab MILWAUKEE, WI.
Adding LA7V HOST XYZ
Updating LAB AUTO INSTRUMENT file (#62.4) for HOST Lab MILWAUKEE, WI.
Adding LA7V HOST XYZ
HL7 v1.6 Environment setup is complete!!
Enter RETURN to continue or '^' to exit:
--------------------------------------------------------------------------HOST Lab Setup
---------------------------------------------------------------------------1.
2.
3.
4.
5.
HOST Lab:
MILWAUKEE, WI (Uneditable)
Logical Link (MailMan or TCP/IP):
Auto Instrument:
Message Configuration: LA7V HOST XYZ
Alert Condition:
Enter a number (1-5): 2
---------------------------------------------------------------------------Logical Link for transmissions to/from MILWAUKEE, WI
----------------------------------------------------------------------------Protocol
Logical Link
-----------------------LA7V Process Results from XYZ
LA7V Send Order to XYZ
Enter a Logical Link:
(MM/TCP): TCP/IP
Updating HL LOGICAL LINK file (#870).
Adding LA7V XYZ
Updating the PROTOCOL file (#101).
68
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
Step #7 continues
---------------------------------------------------------------------------HOST Lab Setup
---------------------------------------------------------------------------1.
2.
3.
4.
5.
HOST Lab:
REGION 7 ISC,TX (DEMO) (Uneditable)
Logical Link (MailMan or TCP/IP): DATA
Automated Instrument:
GRACE PERIOD FOR MESSAGES:
ALERT CONDITION:
Enter a number (1-5): 2
---------------------------------------------------------------------------Logical Link for transmissions to/from REGION 7 ISC,TX (DEMO)
---------------------------------------------------------------------------Protocol
Logical Link
-----------------------LA7V Process Results from 170
LA7V 170 (TCP)
LA7V Send Query to 170
LA7V 170 (TCP)
LA7V Send Order to 170
LA7V 170 (TCP)
Enter a Logical Link:
(MM/TCP): ?
Enter a code from the list.
Select one of the following:
MM
TCP
MAILMAN
TCP/IP
Enter a Logical Link: (MM/TCP): ??
Enter a code from the list.
Select one of the following:
MM
TCP
MAILMAN
TCP/IP
Enter a Logical Link:
February 2002
(MM/TCP): ^
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
69
LEDI II Implementation Instructions
Step #7 continued
---------------------------------------------------------------------------HOST Lab Setup
---------------------------------------------------------------------------1.
2.
3.
4.
5.
HOST Lab:
MILWAUKEE, WI<RET> (Uneditable)
Logical Link (MailMan or TCP/IP): DATA
Auto Instrument:
Message Configuration: LA7V HOST XYZ
Alert Condition:
Enter a number (1-5): 3
AUTOMATED INSTRUMENT: LA7V HOST XYZ
LOAD/WORK LIST: SENDOUTS
METHOD: XYZ REF LAB??
ANSWER MUST BE 1-10 CHARACTERS IN LENGTH
METHOD: XYZ LAB
DEFAULT ACCESSION AREA: SEND SENDOUTS
OVERLAY DATA: Y YES
STORE REMARKS: YES//
Add Chem Tests to the LA7V HOST XYZ Automated Instrument for MILWAUKEE, WI.
Select CHEM TESTS: PSA
1
PSA PROSTATE SPECIFIC ANTIGEN (AXSYM STUDY)
2
PSA-% FREE
3
PSA-TOTAL
CHOOSE 1-3: 1 PROSTATE SPECIFIC ANTIGEN (AXSYM STUDY)
Are you adding 'PROSTATE SPECIFIC ANTIGEN (AXSYM STUDY)' as
a new CHEM TESTS (the 1ST for this AUTO INSTRUMENT)? No// Y
CHEM TESTS NUMBER: 1//
CHEM TESTS TEST: PROSTATE SPECIFIC ANTIGEN (AXSYM STUDY) //
TEST: PROSTATE SPECIFIC ANTIGEN (AXSYM STUDY)//
PARAM 1:
UI TEST CODE: 89760.0000// PSAXYZ
NUMBER OF DECIMAL PLACES: 1
CONVERT RESULT TO REMARK: N NO
ACCEPT RESULTS FOR THIS TEST: Y YES
IGNORE RESULTS NOT ORDERED: Y YES
REMOVE SPACES FROM RESULT: N NO
STORE REMARKS: YES//
YES
REMARK PREFIX: PSA
STORE PRODUCER'S ID: Y YES
STORE REFERENCE RANGE: Y YES
STORE ABNORMAL FLAGS: Y YES
70
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
(Yes)
February 2002
LEDI II Implementation Instructions
Step #7 continued
---------------------------------------------------------------------------HOST Lab Setup
---------------------------------------------------------------------------1. HOST Lab:
XYZ REFERENCE LAB (Uneditable)
2. Logical Link (MailMan or TCP/IP): DATA
3. Auto Instrument: DATA
4. Message Configuration: LA7V HOST XYZ
5. Alert Condition:
Enter a number (1-5): 4
Enter a number (1-5): 4
GRACE PERIOD FOR MESSAGES: 30// ??
Grace period determines the number of days that messages for this
configuration are kept on the system before purging when the message
status is "purgable". If this field is left blank, the system assumes 3
days. These messages are found in the LA7 MESSAGE QUEUE file (#62.49).
When messages have status of "error" they remain on the system until
their corresponding error message is removed from the XTMP global by a
KERNEL cleanup task. The messages then become "purgable".
GRACE PERIOD FOR MESSAGES: 30//
LOG ERRORS: ON// ??
If turned on, errors or exceptional conditions that occur during
message processing are stored in the ^XTMP global for review. To review the
log, in programmer mode, type D PRINT^LA7LOG.
Choose from:
0
OFF
1
ON
LOG ERRORS: ON//
MULTIPLE ORDERS: SINGLE ORDER// ??
Determines when building a HL7 message if message should contain only
one patient/order or multiple patients/orders.
Default is multiple patients per message if possible.
This allows site to configure message building when communicating with
a non-VA system that can not handle a message that has more than one
patient in the message. It applies to both order (ORM) and result
(ORU) messages.
When communicating with a VA facility this field can be left blank
(default) or set to 0 - MULTIPLE PATIENTS
If the receiving system can only accept one patient per HL7 message
then select 1 - SINGLE PATIENT. This will place multiple orders or
results for multiple orders in one message but only one patient will
be contained in the message.
If the receiving system can only accept one order per HL7 message then
select 2 - SINGLE ORDER. This will place in the message one order or
the results associated with one order for a single patient.
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
71
LEDI II Implementation Instructions
Step #7 continued
Note: An order in the VA is considered those tests found on one
accession. What the VA considers as an accession other non-VA systems
may refer to as an order.
Choose from:
0
MULTIPLE PATIENTS
1
SINGLE PATIENT
2
SINGLE ORDER
MULTIPLE ORDERS: 2 SINGLE ORDER//<RET>
---------------------------------------------------------------------------HOST Lab Setup
---------------------------------------------------------------------------1.
2.
3.
4.
5.
HOST Lab:
REGION 7 ISC,TX (DEMO) (Uneditable)
Logical Link (MailMan or TCP/IP): DATA<RET>
Automated Instrument: DATA <RET>
GRACE PERIOD FOR MESSAGES: 4<RET>
ALERT CONDITION:
Enter a number (1-5): 5 <RET>
Select ALERT CONDITION: ? <RET>
You may enter a new ALERT CONDITION, if you wish
Enter "1" to receive alerts for new results, a "2" to receive alerts
for errors during processing. and "3" when orders are received.
Error on message alert may only be selected if Field #4, LOG
ERRORS,is set to "ON".
Choose from:
1
NEW RESULTS
2
ERROR ON MESSAGE
3
ORDERS RECEIVED
Select ALERT CONDITION:NEW (1
NEW RESULTS)<RET>
Are you adding 'NEW RESULTS' as a new ALERT CONDITION (the 1ST for this LA7
MESSAGE PARAMETER)? No//Y (Yes)<RET>
MAIL GROUP:LAB REPORT
72
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
Step #8 IRM: Completing the HL7 Environment Setup
The HL LOGICAL LINK file (#870) contains the links used by the HL7 software to
send messages. This file stores parameters that define the actions of the lower level
protocols and information used within the Systems Link Monitor, which provides the
users feedback regarding the status of each link.
IRM: Use the Link Edit [HL EDIT LOGICAL LINKS] option within the Filer and
Link Management Options [HL MENU FILER LINK MGT] menu within the HL7
Main Menu [HL MAIN MENU] to edit the HL LOGICAL LINK file (#870),
AUTOSTART field (#4.5) in the Logical Link Information section. Set the
AUTOSTART field (#4.5) entry to ‘1’ (Enabled) if you want this to start automatically
after TaskMan is restarted. Otherwise, these links will need to be manually started
using the Start/Stop Links [HL START] option within the Filer and Link
Management Options [HL MENU FILER LINK MGT] menu within the HL7 Main
Menu [HL MAIN MENU].
1a. TCP/IP Interfaces
NOTE: Please consult the VISTA HEALTH LEVEL SEVEN (HL7) SITE
MANAGER & DEVELOPER MANUAL for information on single and multithreaded listeners.
IRM: Complete the HL7 Environment Setup.
To utilize TCP/IP as the communication protocol the following actions are required:
IRM: Coordinate with the facility that will be receiving and sending LEDI HL7
messages. Both sites need to have installed patch HL*1.6*19. Determine the
respective IP addresses and sockets (ports) that both sites will be using to exchange
messages. Sockets in the range 10000-20000 are recommended for the Lab HL7
server (listener) from the sites pool of available sockets. See Port Assignments for
TCP [DBA TCP PORT ASSIGNMENTS] option on FORUM.
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
73
LEDI II Implementation Instructions
Step #8 continues
In the following (i.e.,xxx) is your VA facility number and (i.e., yyy) is the VA facility
number of the system you are exchanging HL7 messages with. In the case of non-VA
facilities yyy will contain the three-letter identifier specified in the LAB SHIPPING
CONFIGURATION file (#62.9), NON-VA SYSTEM IDENTIFIER field (#.11) that
relates to this link.
LA7V xxx (listener) is used to receive order/result messages. All facilities that are
sending order/result messages will connect to this link to transmit LEDI HL7
messages to your facility. This is why this link should be set to PERSISTENT: NO.
Otherwise the link will be tied up and other systems will not be able to connect and
transmit messages to you. The client type links will connect to this listener to
transmit order/result messages. Collection facilities will receive their test result
messages via this link. Host facilities will receive their order messages (shipping
manifest) via this link. If the two systems are host and collection labs for each other,
then both types of messages will be received via this link. This link will ONLY need to
be configured the first time a facility is setting up a TCP/IP connection to another
facility. When setting up additional facilities just provide the client facilities with the
TCP/IP address and port number that this server is using.
LA7V yyy (client) is used to send order/result messages. This is the link that connects
to the other system’s listener (link above) to transmit order/result messages.
Collection facilities will transmit their order messages (shipping manifests) via this
link. Host facilities will transmit their result messages via this link. If the two
systems are both host and collecting labs for each other, then both types of messages
will be sent via this link.
IRM: Provide the TCP/IP address/port that the LA7V xxx server will be listening on
to the clients that will be connecting to your system. You will also need the TCP/IP
address/port of the facility/system you will be transmitting messages to. The other
system must support a non-persistent connection.
The LEDI Setup [LA7V SETUP] option populates the HL LOGICAL LINK file
(#870), NODE field (#.01), and LLP TYPE field (#2).
74
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Implementation Instructions
Step #8 continues
IRM: Edit the following fields within the TCP LLP Parameters section:
On a server logical link these fields:
TCP/IP Address: Null (DSM for OpenVMS); IP address of listener system (Caché for
NT)
TCP/IP Port: Port (socket) that your system is listening on.
Startup Node: Sets the startup node for the listener when the link is Autostarted.
This setting is honored if a) a Task Manager process is running on the specified
startup node, or b) you are an OpenVMS site running TaskMan in a DCL context.
TCP/IP Service Type: Single Listener.
Autostart: If ENABLED, will start up automatically on system boot
On a client logical link the fields:
TCP/IP Address: IP address of target system you are going to connect to.
TCP/IP Port: Port (socket) that target system is listening on.
Persistent: NO
Startup Node: Set the startup node for the link. This setting is honored if a) Task
Manager is running on the specified startup node, or b) you are an OpenVMS site
running TaskMan in a DCL context.
TCP/IP Service Type: Client (Sender).
Autostart: If ENABLED, will start up automatically on system boot.
1b. MailMan Interfaces
No additional setup required.
NOTE: VA MailMan Interfaces utilizes only one Logical Link.
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
75
LEDI II Implementation Instructions
76
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Configuration Setup Checklist
This checklist is provided to ensure that all required LEDI II Implementation Setup
instructions has been completed.
Checklist for Collection Facility setting up Host Facility
NOTE: Use this checklist to confirm that the LEDI II Step #5 LIM
(COLLECTION facility): Lab Shipping Files Setup instructions located in the
Implementation Section of this manual has been completed as instructed.
1. Enter the name of the host facility in the INSTITUTION file (#4), NAME field
(.01). The Institution Master File maintains nationally controlled entries. [ ]
If the host facility is a VA Institution, then verify that the: [ ]
AGENCY CODE field (#95) = “VA” and the
STATION NUMBER field (#99) = A unique Central Office assigned station
number.
If host facility is a non-VA Institution then verify that the: [ ]
AGENCY CODE field (#95) = “OTHER” and the
STATION NUMBER field (#99) = <blank>
2. Setup LAB SHIPPING METHOD file (#62.92). [ ]
Use Edit Shipping Method [LA7S EDIT 62.92] option.
3. Setup LAB SHIPPING CONDITION file (#62.93). [ ]
Use Edit Shipping Condition [LA7S EDIT 62.93] option.
4. Setup LAB SHIPPING CONTAINER file (#62.91). [ ]
Use Edit Shipping Container [LA7S EDIT 62.91] option.
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
77
LEDI II Configruation Setup Checklist
5. Setup LAB SHIPPING CONFIGURATION (#62.9). [ ]
Use the Edit Shipping Configuration [LA7S EDIT 62.9] option to configure this
file. When initially setting up the lab shipping configuration DO NOT edit the
LAB MESSAGING LINK field (#.07) of this file. This field will be filled in after
the LEDI Setup option [LA7V SETUP] configures the HL7 interface for this
configuration. If the host facility is a non-VA facility then determine a three-letter
identifier to assign in lieu of the VA Station Number.
NOTE: See Step #5 LIM (COLLECTION facility): Lab Shipping Files Setup
in the Implementation Section of this manual for examples used during software
testing for several Commercial Reference Laboratories.
6. When setting up an electronic link (HL7 interface) to the host facility laboratory
use the LEDI Setup [LA7V SETUP] option to add/edit a host lab. This option will
setup the Lab and HL7 files for the HL7 interface to the host facility. [ ]
7. If using HL7 TCP/IP interface have IRM complete the HL LOGICAL LINK file
(#870) setup. [ ]
8. If an electronic link (HL7 interface) has been setup for this host facility then use
Edit Shipping Configuration [LA7S EDIT 62.9] option to edit the LAB
MESSAGING LINK field (#.07) to link the shipping configuration to the HL7
interface. Select the message configuration “LA7V HOST xxx” (i.e., xxx is the VA
station number of the host facility or the three-letter identifier previously
assigned for a non-VA facility). The LEDI Setup [LA7V SETUP] option will
attempt to fill in this field. However, if you have multiple shipping configurations
for the same collection and host facility you will need to manually setup these
configurations. [ ]
78
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
LEDI II Configruation Setup Checklist
Checklist for Host Facility setting up Collection Facility
NOTE: Use this checklist to ensure that Step #6 LIM (HOST facility): LAB
SHIPPING CONFIGURATION file (#62.9) Setup located in the Implementation
section of this manual has been completed.
1. Enter/configure the collecting facility entry in INSTITUTION file (#4). The
Institution Master File maintains nationally controlled entries. [ ]
If the collecting facility is a VA institution then verify that: [ ]
AGENCY CODE field (#95) = “VA”
STATION NUMBER field (#99) = Unique Central Office assigned station
number.
If the collecting facility is a non-VA institution then verify that: [ ]
AGENCY CODE field (#95) = “OTHER”
STATION NUMBER field (#99) = <blank>
2. Setup SHIPPING CONFIGURATION file (#62.9). [ ]
Use Edit Shipping Configuration [LA7S EDIT 62.9] option.
Specify, “Host facility”, when asked how the configuration is being edited.
Add local lab tests that the Collecting facility will be shipping, specifiying local
collection sample and urgency to use to build the LAB PENDING ORDERS
ENTRY file (#69.6) entries. If collecting facility is a non-VA facility then
determine a three-letter identifier to assign in lieu of the VA Station Number. See
Step #5 LIM: Lab Shipping Files Setup above for suggested values of some
commercial reference laboratories.
3. If setting up an electronic link (HL7 interface) to the collection lab use LEDI
Setup [LA7V SETUP] option to add/edit a collection lab. This option will setup the
Lab and HL7 files for the HL7 interface to the collection facility. [ ]
4. If using HL7 TCP/IP interface then have IRM complete the HL LOGICAL LINK
file (#870) setup. [ ]
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
79
LEDI II Configruation Setup Checklist
80
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
Package Operation
This section contains information regarding the VISTA LEDI II Installation Guide
and User Manual retrival formats and on-line access locations. The LEDI II new and
modified menus and options descriptions are also included in this section.
LEDI II Documentation Retrieval Formats
LEDI II Installation Guide and User Manual files are exported in the following
formats:
File Names
Contents
Retrieval Formats
================
===================
==================
LAB_LEDI_II_IG.PDF LABORATORY ELECTRONIC DATA
BINARY
LAB_LEDI_II_UM.PDF LABORATORY ELECTRONIC DATA
BINARY
LEDI II Documentation Retrieval Locations
NOTE: All sites are encouraged to use the File Transfer Protocol (FTP) capability.
Use the FTP address “download.vista.med.va.gov” (without the quotes) to connect to
the first available FTP server where the files are located.
LEDI II Installation Guide and User Manual files are available on the
ANONYMOUS.SOFTWARE directories at the following Office of Information Field
Offices (OIFOs): (LAB_LEDI_II_IG.PDF and LAB_LEDI_II_UM.PDF).
OI FIELD OFFICE
FTP ADDRESS
================
================
========================
ALBANY
ftp.fo-albany.med.va.gov
HINES
ftp.fo-hines.med.va.gov
SALT LAKE
ftp.fo-slc.med.va.gov
February 2002
DIRECTORY
[ANONYMOUS.SOFTWARE]
[ANONYMOUS.SOFTWARE]
[ANONYMOUS.SOFTWARE]
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
81
Package Operation
Website Locations:
The VISTA LEDI II Installation Guide (i.e., LAB_LEDI_II_IG.PDF and
LAB_LEDI_II_IG.DOC) and LEDI II User Manual (i.e., LAB_LEDI_II_UM.PDF
and LAB_LEDI_II_UM.DOC) in Portable Document Format (PDF) and MS Word
(DOC) Format are available at the following VISTA Intranet locations:
Laboratory Version 5.2 Home Page:
http://vista.med.va.gov/ClinicalSpecialties/lab/
VISTA Documentation Library (VDL):
http://vista.med.va.gov/vdl/
82
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
Package Operation
LEDI II New and Modified Options
The following options were created or modified to accommodate LEDI II new
functionality:
LEDI II New Options
Edit Required Test Information [LA7S MANIFEST TEST REQ INFO]
option
This option allows a user to enter/edit information that is required to be sent with a
Lab test when it is shipped. Examples can be the total volume of urine collected for a
timed urine test, weight of specimen collected, duration of specimen collection,
patient height, and weight.
Retransmit LEDI Lab Results [LA7S RESULTS RETRANSMIT] option:
This new option allows a user at a host facility to select one or more accessions that
were received from other LEDI collection facility and retransmit test results
associated with the accession(s) to the sending/collecting facility via HL7 messaging.
These two new options are located on the Lab Shipping Menu [LA7S MAIN MENU],
which is located on the Laboratory DHCP Menu [LRMENU].
LEDI II Modified Options
Print LEDI Pending Orders [LA7S PENDING PRINT LEDI] option:
This option is used by the host facility to print a collection facility LEDI shipping
manifest report from the host facility's LAB PENDING ORDERS ENTRY file (#69.6).
This option can be used to reprint a shipping manifest report, which is lost or
damaged during shipping. The user is prompted to enter the shipping manifest
number, which prints a barcoded-shipping manifest report containing all of the
patients entered for that manifest. The new shipping manifest report format is
similiar to the regular shipping manifest report received from the collection
(shipping) facility.
Display Lab Universal Interface Message [LA7 PRINT LAB UI MESSAGE]
option:
The logic used to print error messages associated with a UI message is modified to
prompt the user when the Kernel Browser should be used to display a form message.
If the browser is not selected then a standard “scroll and roll” display of the selected
message(s) is produced. This option is located on the Lab Universal Interface Menu
[LA7 MAIN MENU]. This submenu is located on the Lab Interface Menu [LA
INTERFACE].
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
83
Package Operation
LEDI II Menus and Options
LEDI II software application contains two menus; the Lab Shipping Management
Menu [LA7S MGR MENU] is assigned to the LIM, and Lab Shipping Menu [LA7S
MAIN MENU]) is assigned to the user. The Lab Universal Interface Menu [LA7
MAIN MENU] and Accessioning Menu [LR IN] are also exported with this release.
The following charts contain detailed descriptions of all menus and options exported
by this release:
Lab Shipping Management Menu [LA7S MGR MENU]
The Lab Shipping Management menu [LA7S MGR MENU] contains all
implementation related options available for the LIM.
LEDI Menu
Options
Lab Shipping
Management Menu
[LA7S MGR MENU]
Edit Shipping
Configuration [LA7S
EDIT 62.9] option
Edit Shipping
Container [LA7S
EDIT 62.91] option
Edit Shipping Method
[LA7S EDIT 62.92]
option
Edit Shipping
Conditions [LA7S
EDIT 62.93] option
LEDI Setup [LA7V
SETUP] option
84
Description
This submenu can be found on the Lab liaison menu
[LRLIAISON] containing the following options:
This option sets up the LAB SHIPPING CONFIGURATION
file (#62.9).
See example in the Implementation section Step #5.
This option sets up the LAB SHIPPING CONTAINER file
(#62.91).
See example in the Implementation section Step #5.
This option sets up the LAB SHIPPING METHOD file
(#62.92).
See example in the Implementation section Step #5.
This option sets up the LAB SHIPPING CONDITIONS file
(#62.93).
See example in the Implementation section Step #5.
This option sets up the HL7 and Lab Auto Instrument
environment for a HOST or COLLECTION system. This
option also sets up entries in the:
• HL7 APPLICATION PARAMETER file (#771)
• HL LOGICAL LINK file (#870)
• HL LOWER LEVEL PROTOCOL PARAMETER file
(#869.2)
• PROTOCOL file (#101)
• AUTO INSTRUMENT file (#62.4)
• LA7 MESSAGE PARAMETER file (#62.48)
See examples in the Implementation section Steps #6 and #7.
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
Package Operation
Electronic Catalog Menu [LA7S CATALOG MENU]
This menu contains the options that maintain, produce, and print the laboratory test
available for the collection site to order.
LEDI Menu
Options
Electronic Catalog
Menu [LA7S
CATALOG MENU]
Electronic Catalog
Information Entry
[LA7S CATALOG
ENTRY] option
View Individual
Electronic Catalog
Entry [LA7S VIEW
INDIVIDUAL
ENTRY] option
Electronic Catalog
Print [LA7S PRINT
CATALOG] option
February 2002
Description
This submenu is located on the Lab Shipping Management
Menu [LA7S MGR MENU] consisting of the following
options:
This option allows the user to enter laboratory test
information for the electronic catalog.
For an example, see the Option Examples section.
This option allows the user to view or print an individual
entry in the Electronic Test Catalog.
For an example, see the Option Examples section.
This option allows the user to produce a report of all catalog
entries.
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
85
Package Operation
Lab Shipping Menu [LA7S MAIN MENU]
The Lab Shipping [LA7S MAIN MENU] Menu contains 10 options, including two
new options (i.e., Retransmit LEDI Lab Results [LA7S RESULTS RETRANSMIT]
and Edit Required Test Information [LA7 MANIFEST TEST REQ INFO]). The
following charts include a detailed description of the existing LEDI options and the
two new LEDI II options:
LEDI Menu
Options
Lab Shipping
Menu [LA7S MAIN
MENU]
Add/Remove a
Shipping Manifest
[LA7S MANIFEST
TEST ADD/
REMOVE] option
Build Shipping
Manifest [LA7S
MANIFEST BUILD]
option
Cancel a Shipping
Manifest [LA7S
MANIFEST
CANCEL] option
Close/Ship a
Shipping Manifest
[LA7S MANIFEST
CLOSE/SHIP] option
Edit Required Test
Information [LA7
MANIFEST TEST
REQ INFO] option
86
Descriptions
The Lab Shipping Menu [LA7S MAIN MENU] contains all
the workload related options available to the LIM. This
submenu is located on the Laboratory DHCP [LRMENU]
menu consisting of the following 10 options:
This option allows a test to be added or removed from an
“Open” shipping manifest.
This option builds specimens on to a shipping manifest.
This option cancels an open shipping manifest. All tests are
removed and made available for building on another
shipping manifest.
This option marks a shipping manifest as closed and/or
shipped.
This new option allows a user to enter/edit information
required to be sent with a lab test when it is shipped.
Examples can be the total volume of urine collected for a
timed urine test, weight of specimen collected, duration of
specimen collection, patient height and weight.
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
Package Operation
Lab Shipping [LA7S MAIN MENU] Menu continued
LEDI Menu
Options
Order Status Report
[LA7S ORDER
STATUS REPORT]
option
Print LEDI Pending
Orders [LA7S
PENDING PRINT
LEDI] option
Print Shipping
Manifest [LA7S
MANIFEST PRINT]
option
Retransmit LEDI
Lab Results [LA7S
RESULTS
RETRANSMIT]
option
Retransmit Shipping
Manifest [LA7S
MANIFEST
RETRANSMIT]
option
February 2002
Descriptions
This report allows the user to produce a report based on
selected criteria.
This option is used to print a collecting facility’s LEDI
shipping manifest from the host facility’s LAB PENDING
ORDER ENTRY (#69.6) file. This option could be used to
reprint a shipping manifest lost or damaged during
shipment. The user is prompted for the shipping manifest
number. The option will then print a barcoded-shipping
manifest containing all of the patients on that manifest. The
format is similar to the regular shipping manifest received
from the collecting (shipping) facility.
This option allows the user to print a shipping manifest list
for lab specimens sent outside the facility to a reference lab.
This new option allows a user at a host facility to select one
or more accessions that were received from other LEDI
collecting facilities and retransmit results associated with
the accession(s) to the sending/collecting facility via HL7
messaging.
This option allows the users to retransmit a shipping
manifest electronically to the host facility. Manifest needs to
be in status of “SHIPPED” to be able to select and the
shipping configuration associated with the manifest must
be “ACTIVE”.
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
87
Package Operation
Lab Messaging Nightly Cleanup [LA7TASK NIGHTY] option
This is a stand-alone option that should be scheduled to run daily via TaskMan as a
scheduled task.
Options
Lab Messaging
Nightly Cleanup
[LA7TASK NIGHTY]
option
Description
This is tasked option is used to check the integrity of the
LA7 MESSAGE QUEUE file (#62.49), and to purge
messages that are eligible for purging.
This option should be tasked daily, preferably during a
period when activity in the Lab Messaging (that is,
Universal Interface, LEDI) package is at a minimum.
Before the purge of LA7 MESSAGE QUEUE file (#62.49),
an integrity check is performed. The integrity check can be
run with a couple of switches.
LA7FIX = 0 - do not fix
1 - do fix errors
LA7LOG = 0 - do not log
1 - do log errors in
LA7ION = name of device
log errors (LA7LOG=1)
errors
errors in XTMP global.
XTMP global
to print error report if set to
These parameters can be setup by TaskMan if the site
defines them when scheduling the task.
Example:
Edit Option Schedule Option Name: LA7TASK NIGHTY
VARIABLE NAME: LA7FIX VALUE: 0
VARIABLE NAME: LA7ION VALUE: "IRM DEVELOP LASER1"
VARIABLE NAME: LA7LOG VALUE: 1
If errors are found, an alert is sent to members of the mail
group “LAB MESSAGING” notifying them that errors were
detected. If logging of errors occurred then alert recipients
will be able to print or view error log from the alert system.
Alternatively the error report can be printed using the Print
Lab Messaging Integrity Check Report [LA7 PRINT
INTEGRITY CHECK] option. The integrity report can be
run alone using Lab Messaging File Integrity Checker [LA7
CHECK FILES] option.
For an example, see the Implementation section Step #3.
88
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
Package Operation
Lab Universal Interface (LA7 MAIN MENU) Menu
The Lab Universal Interface Menu [LA7 MAIN MENU] contains one modified
option for the release of LEDI II. This menu is located on the Lab Interface Menu
[LA INTERFACE]:
LAB UI Menu
Options
Lab Universal
Interface Menu
(LA7 MAIN MENU)
Display Lab UI
Message [LA7
PRINT LAB UI
MESSAGE] option
Download to UI [LA7
ADL SEND] option
Lab Messaging File
Integrity Checker
[LA7 CHECK
FILES] option
Print Lab Messaging
Integrity Check
Report [LA7 PRINT
INTEGRITY
CHECK] option
Start/Stop Auto
Download
Background Job
[LA7 ADL
START/STOP]
option:
February 2002
Descriptions
The Lab Universal Interface Main Menu contains options to
setup, manage and use the Lab Universal Interface. This
submenu is located on the Lab interface Menu [LA
INTERFACE] containing one modified option:
This option logic used to print error messages associated
with a UI message is modified to prompt the user when the
Kernel Browser should be used to display a message. If the
browser is not selected then a standard “scroll and roll”
display of the selected message(s) is produced.
This option fixes the logic to handle alphanumeric UIDs
when building listing of accession to schedule for
downloading.
This option performs the following checks:
*Integrity of LA7 MESSAGE QUEUE file (#62.49).
*Entries having proper cross-references and crossreferences pointing to the correct entry.
*Fixes bad entries.
*Provides a printed report of errors found.
This option prints a Lab Messaging Integrity Check Report.
This option adds a display of the current number of
accessions awaiting checks to determine if the accession
meets the criteria for downloading to an instrument.
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
89
Package Operation
Accessioning [LR IN] Menu
The Accessioning menu [LR IN] is located on the Laboratory DHCP menu
[LRMENU].
LEDI Menu
Options
Accessioning menu
[LR IN]
Referral Patient
Multi-purpose
Accession [LRLEDI]
option
Descriptions
This submenu is located on the Laboratory DHCP menu
[LRMENU] and contains the following option:
This option is used to accession referral patients from
another medical center. The patient must already exist in
the PATIENT file (#2) to be selected. The option will
accession the patient into the REFERRAL PATIENT file
(#67) if it does not already exist.
The user will be prompted for the Unique Identifier. The
user will then be able to order tests for that patient.
For an example, see the Option Examples section.
90
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
Package Operation
LEDI Menu and Option Examples
Electronic Catalog Information Entry [LA7S CATALOG ENTRY]
This option allows the user to enter laboratory test information for the electronic
catalog.
Example: ENT Electronic Catalog Information Entry option
ENT
IND
ALL
Electronic Catalog Information Entry
View Individual Electronic Catalog Entry
Electronic Catalog Print
Select Electronic Catalog Menu Option: ENT
Entry
Electronic Catalog Information
Select LABORATORY TEST NAME:
PROSTATE SPECIFIC ANTIGEN
NAME: PROSTATE SPECIFIC ANTIGEN Replace
NATIONAL VA LAB CODE: Prostate Specific Ag//<RET>
RESULT NLT CODE: Prostate Specific Ag//<RET>
CATALOG ITEM: YES//<RET>
COST: 11.18
PRICE: 22.36
Select SPECIMEN: SERUM//<RET>
SPECIMEN: SERUM//<RET>
SP COST: 21.32
SP PRICE: 32.72
Select SPECIMEN: ^
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
91
Package Operation
View Individual Electronic Catalog Entry [LA7S VIEW INDIVIDUAL
ENTRY]
This option allows the user to view or print an individual entry in the Electronic Test
Catalog.
Example: IND View Individual Electronic Catalog Entry option
ENT
IND
ALL
Electronic Catalog Information Entry
View Individual Electronic Catalog Entry
Electronic Catalog Print
Select Electronic Catalog Menu Option: IND
Catalog Entry
Select LABORATORY TEST NAME:
DEVICE:
View Individual Electronic
PROSTATE SPECIFIC ANTIGEN
VIRTUAL TERMINAL
Electronic Test Catalog
JUL 10,1997 14:08
PAGE 1
-----------------------------------------------------------------------PROSTATE SPECIFIC ANTIGEN
Collection Tube: BLOOD
RED OR GOLD
Ship 1 ml aliquot; Refrigerate
Site/Specimen: SERUM
Snomed: 0X500
Reference Low: 0
Reference High: 4.0
Units: ng/mL
92
NLT Code: 89760.0000
NLT Result Code: 89760.0000
+Prostate Specific Ag
+Prostate Specific Ag
Default Cost: 11.18
Specimen: SERUM
Specimen Cost: 21.32
Default Price: 22.36
Specimen Price: 32.72
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
Package Operation
Referral Patient Multi-purpose Accession [LRLEDI]
The Host facility laboratory users must record all patient’s demographics and
accession the specimens received from the Collection facility laboratory. The Edit
Shipping Configuration option is used for recording this data.
The Referral Patient Multi-purpose Accession [LRLEDI] option prompts the user to
respond to the question “are you using a bar-code reader?” the user may accept the
default of “YES” or type “NO” as a response.
When the default of “YES” (“are you using a bar-code reader?”) is accepted for the
Shipping Configuration the user may scan the bar coded Shipping Manifest list
(initiated by the Collection facility laboratory). The bar coded Shipping Manifest list
contains the patient’s demographics and specimens accessioning data. The Shipping
Manifest data are stored in the LAB SHIPPING MANIFEST file (#62.8) or the LAB
PENDING ORDERS ENTRY file (#62.9) at the Collection facility.
The selection not to use a bar code reader requires the users to type in the patient’s
demographics (i.e., name or social security number) before the specimens can be
accessioned. This selection allows typographical mistakes and the possibility of
duplicate entries.
Example 1: Manual Accession
Select Accessioning menu Option: REF
Accession
Referral Patient Multi-purpose
Are you using a barcode reader? YES//NO (No)<RET>
Select Shipping Configuration: WHITE RIVER JUNCTION VAMC<RET>
WANT TO ENTER COLLECTION TIMES? Y//<RET> (Yes)
Select ACCESSION TEST GROUP:CHEMISTRY<RET>
Select Patient Name -'^M' To enter New Name:G4451<RET>
Searching for a Patient file entry
Searching for a Referral Patient
04-25-65
017784451
...OK? Yes//<RET> (Yes)
NAME: GREAT,JOY
SEX: FEMALE
GREAT,JOY
FEMALE
DOB: 04/25/65
IDENTIFIER:017784451<RET>
Is this the correct patient?YES<RET>
February 2002
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
93
Package Operation
Enter Remote UID: 5250710001
[NOTE: You must manually enter the
COLLECTION LAB UI Number] LAB Order number: 185<RET>
Choose one (or more, separated by commas) ('*' AFTER NUMBER TO CHANGE
URGENCY)
1
ASTRA
23 MAGNESIUM
2
IDEAL
24 OSMOLARITY
3
ELECTROLYTES
25 PO4
4
ACID PHOSPHATASE
26 POTASSIUM
5
ALKALINE PHOSPHATASE
27 PROTEIN,TOTAL
6
ALBUMIN
28 PROTEIN ELECTROPHORESIS
7
AMYLASE
29 SALICYLATE
8
CALCIUM
30 SGOT
9
CHLORIDE
31 SGPT
10 CHOLESTEROL
32 SODIUM
11 CO2
33 THEOPHYLLINE
12 CPK
34 TOT. BILIRUBIN
13 CPK ISOENZYMES
35 TRIGLYCERIDE
14 CREATININE
36 UREA NITROGEN
15 DIR. BILIRUBIN
37 URIC ACID
16 ETHANOL
38 PROTEIN (CSF)
17 GENTAMICIN
39 LIPID PROFILE
18 GENTAMICIN (PRE)
40 CARDIAC ENZYMES (SGOT,LDH,CPK)
19 GENTAMICIN (POST)
41 LFTS
20 GLUCOSE
42 ARTHRITIS PROFILE
21 LDH
43 CX-3A:NA,K,CL,CO2,BUN,CREA,GLU
22 LDH ISOENZYMES
44 CX-3B:LYTES,BUN,CREA,GLU,CA
TEST number(s): 8
Other tests? N// Y (Yes)
You have just selected the following tests for GREAT,JOY 017-78-4451
entry no. Test
Sample
1
CALCIUM
BLOOD SERUM
All satisfactory? Yes// <RET>
(Yes)
LAB Order number: 185
Collection Date@Time: NOW// <RET> (JUL 10, 1997@10:56:30)
PRINT LABELS ON: LABEL PRINTER
printer]
[NOTE: Enter the name of your site’s label
DO YOU WISH TO TEST THE LABEL PRINTER? No//<RET>
ACCESSION:
CALCIUM
R/CH 0710 17
(No)
<5250710001>
BLOOD SERUM
Select Patient Name -'^M' To enter New Name: ^
94
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
Package Operation
Example 2: Using Bar Code Reader
Select Accessioning menu Option:REF<RET>
Accession
Referral Patient Multi-purpose
Are you using a barcode reader? YES//<RET>(Yes)
Scan Remote Site Bar code (SM):<RET>
STX^SITE^521^2970623.133509^521-19970623-2^ETX
[NOTE: This response appears after the bar code is scanned]
Select ACCESSION TEST GROUP:
CHEMISTRY
Scan Patient/Accession Barcode (PD):<RET>
STX^PD^382486204^521^3471740016^m^2970623.133509^ETX
[NOTE: This response appears after the bar code is scanned]
NAME: FORD,HARRISON
DOB: 04/01/60
SEX: MALE
IDENTIFIER: 000000000
LAB Order number: 201
Choose one (or more, separated by commas)
URGENCY)
1
2
3
4
5
6
7
8
9
10
11
12
13
CHEM 7
CHEM 20
URINALYSIS
LIVER FUNCTION
MAGNESIUM
PO4
LDH
ETHANOL
CPK
CHOLESTEROL
TRIGLYCERIDE
ACID PHOSPHATASE
CALCIUM, URINE
14
15
16
17
18
19
20
21
22
23
24
25
('*' AFTER NUMBER TO CHANGE
CHEM 7, URINE
LIPASE
LITHIUM
AMMONIA
OSMOLARITY
OSMOLARITY, URINE
PROTEIN,TOTAL
LACTIC ACID
HDL
CO2
SODIUM
GLUCOSE
You have just selected the following tests for ROGERS,ROY 100-10-1000
entry no. Test
Sample
1
MAGNESIUM
BLOOD
SERUM
2
GLUCOSE
BLOOD
SERUM
Other tests? N// <RET> (No)
You have just selected the following tests for ROGERS,ROY 100-10-0000
entry no. Test
Sample
1
MAGNESIUM
BLOOD
SERUM
2
GLUCOSE
BLOOD
SERUM
All satisfactory? Yes// <RET>
February 2002
(Yes)
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
95
Package Operation
Example 2: Using Bar Code Reader (continues)
LAB Order number: 201
PRINT LABELS ON: LABLABEL// <RET>
R7 GP
DO YOU WISH TO TEST THE LABEL PRINTER? No// <RET> (No)
~For Test: CPK ISOENZYMES PANEL (IMMONO) WR
BLOOD
SERUM
Enter Order Comment: FIRST SET OF ISO’S SENT
(~FIRST SET OF ISO’S SENT)
OK? Yes// <RET> (Yes)
ACCESSION:
CPK
R/CH 0716 397 <1171970397>
BLOOD
SERUM
ACCESSION: R/SPC 0716 49
TOTAL CK FOR ISO
CPK MB (NG/ML)
CPK MB INDEX
<8671970049>
BLOOD
BLOOD
BLOOD
SERUM
SERUM
SERUM
LAB PROCESSING INSTRUCTIONS: Please freeze sample [NOTE: This comment is from
File #60]
GLUCOSE
GENERAL PROCESSING INST.:
BLOOD
SERUM
Send sample on ice
Scan Patient/Accession Barcode (PD): <RET>
96
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
Package Operation
Turnaround times By Urgency [LR TAT URGENCY]
This option generates a report of the turnaround times for selected lab test for
selected urgencies.
Example: Turnaround times by Urgency
Select Lab statistics menu Option: TURnaround times By Urgency
LEDI Utility - Urgency Turnaround Time
This option generates a report of the turnaround time for selected
lab tests. Enter only those urgencies you want extracted. WKLD urgencies
will be included for each normal urgency selected. Enter the
test(s) you want the report display.
A detailed report is available to show the data being used to
compute the turnaround times.
Regular hours are from 7:01 AM to 5:00 PM
Irregular hours includes all other times, holidays and weekends.
Date to START with: TODAY// <RET> (JUL 16, 1997)
Date to END with: 07/16/97// T (JUL 16, 1997)
Select the laboratory tests to be used in this report -LABORATORY TEST:
GLUCOSE
LABORATORY TEST: <RET>
Urgencies:
STAT
PLEASE DO NOT USE
ROUTINE
Enter all urgencies you want extracted.
URGENCY: STAT
URGENCY: <RET>
Include a detailed report? No// <RET>
DEVICE: HOME//
February 2002
(No)
PRINTER [NOTE: Enter the site’s printer name]
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
97
Package Operation
Example: Turnaround times by Urgency (continues)
LEDI Management Report - Urgency Turnaround Time
From JUL 16,1997 To JUL 16,1997
Date Printed: JUL 16,1997@11:25:26
__________________________________________________________________________
Turnaround Time (TAT) - Regular (0701-1700) hours
Number of tests
---------------------2
Total time
---------62 min
Ave TAT
------31 min
Turnaround Time (TAT) - Irregular hours
Number of tests
---------------------13
Urgencies:
STAT
WKL - STAT
Total time
---------300 min
Ave TAT
------23 min
Tests:
GLUCOSE
98
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
Glossary
The glossary of terms is related to the LEDI II software application:
CAP:
College of American Pathologists
Collection facility:
The laboratory that collects the patient’s
specimen. After the specimen is collected,
the Collection facility should:
*Create an electronic Shipping Manifest
(specifying the lab tests to be performed
by the Host facility laboratory).
*Transmit the Shipping Manifest List to
the Host facility laboratory.
*Transport the specimen by carrier to the
Host facility laboratory.
Data Fields:
The information base associated with a
segment.
Data Type (DT):
Restrictions on the contents of the data
field as defined by the HL7 Standard.
Electronic Catalog:
The new Electronic Catalog contains
laboratory test available for the collection
site to order.
Element Name:
Globally unique descriptive name for the
field.
Host facility:
February 2002
The laboratory that receives the patient’s
specimen and the electronic Shipping
Manifest List (transmitted by the
Collection facility laboratory)
The Host facility performs the following
steps:
*Bar codes the specimen arrival.
*Performs the specified lab test.
*Verifies the lab test results.
*Transmits the lab test results back to
the Collection facility.
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
99
Glossary
LEDI:
Laboratory Electronic Data Interchange
Length (LEN):
The maximum number of characters that
one occurrence of the data field may
occupy.
NLT:
National Laboratory Test
Optionally (R/O/C):
Whether the data field is required,
optional, or conditional in a segment. The
designations are:
*R - required
*O (null) - optional
*C - conditional on the trigger event
PID:
Patient Identification
Repetition (RP/#):
Whether the field may repeat. The
designations are:
N (null) - for no repetition allowed; Y - the
field may (null) - for no repetition
allowed; Y - the field may repeat an
indefinite or site determined number of
times; and (integer) - the field may repeat
up to the number of times specified in the
integer. The ordinal position of the data
field within.
Segments:
A logical grouping of data fields.
Shipping Manifest:
The shipping manifest is a printout list of
lab specimens sent outside the facility to
a reference lab for processing.
Sequence Number (SEQ):
The ordinal position of the data field
within the segment. This number is used
to refer to the data field in the text
comments that follow the segment
definition table.
100
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
February 2002
Glossary
Table (TBL#):
This is a table of values that may be
defined by HL7 or negotiated between
the V IST A Laboratory application and
the vendor system.
TCP/IP:
Transmission Communication
Protocol/Internet Protocol
Trading Partners:
An established relationship between two
or more laboratories for receiving and
processing lab specimens.
UI:
Universal Interface
UID:
Unique Identifier
VAMC:
Department of Veterans Affairs Medical
Center
VISN:
Veterans Integrated Service Network
Veterans Health Information Systems
and Technology Architecture
VISTA Laboratory UI and LEDI HL7 This document specifies an interface to
Interface Standard Specifications V. the VISTA Laboratory software
1.1:
application based upon the Health Level
Seven (HL7) Standard. This interface
forms the basis for the exchange of
healthcare information between the
VISTA Laboratory software application
and all non- VISTA systems. Especially
those non- VISTA systems that generate
laboratory result information.
VISTA
Workload System:
February 2002
This is a method of tracking number of
LEDI specimens processed.
LEDI II User Manual V. 5.2
LA*5.2*46/LR*5.2*222
101