Download User Guide - HSCIC Groups
Transcript
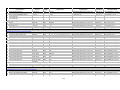
ACE inhibitor therapy for patients with myocardial infarction Purpose: To help reduce the level of risk to health for NHS patients with myocardial infarction (MI) and ensure high standards of primary health care and treatment delivered to them. A number of trials have shown reduced mortality following MI with the use of ACE inhibitors that are also of benefit in reducing adverse coronary events and progression of coronary arteriosclerosis without left ventricular systolic dysfunction. There is evidence that angiotensin II antagonists have a similar effect. Definition of indicator and its variants: Proportion of patients with a history of MI who are treated with an ACE inhibitor or angiotensin II antagonist, unless contraindications or side-effects are recorded. Statistic Sex Age group Organisation (see glossary) Number and percent of patients P All Ages E, GOR, SHA, PCO Period Current data FY 2009/10 File_worksheet Name Trend data 09O_607PC_10_V1 Numerator: Numerator data - Patients with a history of MI diagnosed after 1st April 2003 who are currently (measured as a prescription within the 6 months before the reference date) treated with an ACE inhibitor or angiotensin II antagonist, unless contraindications or side-effects are recorded. Source of numerator data - Quality and Outcomes Framework (QOF). Comments on numerator data - The data for this primary care indicator are derived from the Quality Management Analysis System (QMAS) and supplied by the Prescribing Support Unit at The NHS Information Centre for health and social care. The QOF is part of the General Medical Services (GMS) contract, with a focus on prevalence and management of common chronic diseases at general practice level. It is aimed at measuring the achievement of general practices against a set of evidence based negotiated standards and encouraging good practice. The data to support the clinical quality indicators (i.e. clinical achievement data) are extracted from individual GP practice clinical systems and sent automatically to QMAS once a month. Practices can make additional submissions of clinical data at any time. The ‘reference date’ is the specified date of patient records held by the general practice modelled for the end of the st QOF year (midnight 31 March). It is the same for the data from all practices and may differ from the date of data submission. Denominator: Denominator data - All patients dignosed with MI excluding those who have been exception reported. Patients may be exception reported for the following reasons: st • MI diagnosis recorded before 1 April 2003; • registered in the three months before the reference date; • CHD exception reported in the 15 months before the reference date; • MI diagnosis recorded in the three months before the reference date; • persistent and expiring ACE inhibitor contraindications recorded; • persistent and expiring A2 antagonist contraindications recorded. For more details regarding the description of the selection criteria and dataset rules of the relevant indicator set see the following documentation available at: http://www.wmrlmc.co.uk/gms2/informationtechnology.htm Source of denominator data - See “Numerator data”. Comments on denominator data - The denominator is a count of patients for whom the clinical indicator should be met. However, the selection rules do not provide the same opportunity for all patients included in the denominator to take advantage of the full period (maximum number of months after the diagnosis) allowed by good practice for the test, measurement or treatment to be undertaken (e.g. if good practice for angina patients allows for a particular test to be conducted up to 12 months after diagnosis then patients diagnosed between four and 11 months before the reference date do not have the full period of 12 months for the test). Additionally, patients can be removed from the denominator through the application of exception reporting. 379
Related documents

User Guide - HSCIC Groups

A user's guide to data collected in primary care in England

CORRESPONDENCIAS ENTRE CIE-10 Y CIE

User Guide: Local Alcohol Profiles for England 2014

EasyAudit User Manual - Woodward Associates (UK)

Basic tutorial to CPMD calculations

2 - Sony

HTM 66 covers - Health in Wales

Getting to grips with hoisting

CONTROLS AND INSTRUMENTS 23

User Manual for Taxonomic Editor Version 4

Primary Care Commissioning Application User Guide for v88